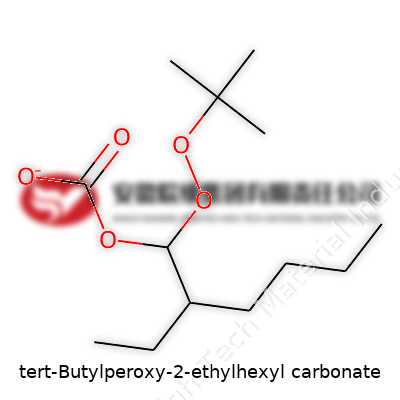
tert-Butylperoxy-2-ethylhexyl Carbonate: A Practical Look at Its Journey and Impact
Historical Development
tert-Butylperoxy-2-ethylhexyl carbonate started showing up in chemical catalogs in the late part of the twentieth century, coming on the scene as industries looked for new ways to initiate polymerization at lower temperatures. Early research grew out of a wave of interest in organic peroxides spurred by the search for safer, more stable free radical sources. Researchers kept tinkering with the carbonate backbone and peroxy moiety, pressing ahead for molecules that could offer both reactivity and controlled decomposition. Over the years, as concerns about workplace accidents and storage hazards grew, chemists zeroed in on tert-butylperoxy-2-ethylhexyl carbonate’s unique blend of activity and safety. Regulatory changes in the 1990s and innovations in peroxide manufacturing helped move production out of the lab and into large-scale plants across North America, Europe, and East Asia. Direct testimonies from plant managers highlight a learning curve, as operators dealt with the challenges of temperature sensitivity and shelf-life management, leading to present-day protocols that keep accidents rare.
Product Overview
tert-Butylperoxy-2-ethylhexyl carbonate comes together as a colorless to pale yellow liquid, packing a punch as a free radical initiator in polymer chemistry. It stands out among organic peroxides, thanks to a balance between robust reactivity and lower handling risks. This compound often goes into the manufacture of plastics and synthetic rubber, lending a reliable mechanism for initiating chain reactions at moderate temperatures, which helps save on energy and boosts yield reliability. Chemical manufacturers rely on its predictable breakdown profile, cutting down waste and unwanted side reactions. Its popularity in both research and factory settings points to practical qualities and a track record of dependable performance in polymerization and modification steps.
Physical & Chemical Properties
The physical makeup of tert-butylperoxy-2-ethylhexyl carbonate gives it a practical charm. With a molecular formula of C13H26O4 and a molecular weight around 246.3 g/mol, it carries the tell-tale signature of organic peroxides: a tendency to decompose at higher temperatures, but offering a relatively high boiling point compared to other peroxy carbonates. Storage calls for cool, shaded environments as the peroxide bond is sensitive to heat, shock, and contamination. Density falls in the range of 0.90 to 0.95 g/cm³. As for solubility, water isn’t much of a friend—organic solvents like ether or nonpolar hydrocarbon bases offer better matches, a feature that lines up well with most industrial applications. Shelf life and purity tests depend heavily on keeping this compound away from catalytic metals, which can trigger unwanted breakdown and pose safety concerns. The telltale peroxide odor, noticeable even at low concentrations, cues workers to handle it with respect.
Technical Specifications & Labeling
Quality standards require manufacturers to list precise composition percentages, stability under various storage and transport conditions, and clear batch-tracking details. Typical certificates of analysis include active oxygen content (measured in percentage), water content, and common impurities like tert-butanol. Some labels use hazard pictograms under GHS standards, warning of both fire and health hazards. The UN number 3109 flags its role as an organic peroxide type F, which mandates routine checks and restricted shipping conditions. Container labeling puts heavy emphasis on both the Reid vapor pressure and maximum temperature thresholds to keep this material from causing incidents during long-term storage or transfer. Technical datasheets give detailed instructions about shelf life, transport restrictions, and protocols in the event of leaks or spills, reflecting hard-learned lessons from decades of work with these peroxides.
Preparation Method
Chemists typically build tert-butylperoxy-2-ethylhexyl carbonate in a two-step reaction, starting from 2-ethylhexan-1-ol and treating it with phosgene or a safer alternative to create 2-ethylhexyl chloroformate. Next, a reaction with tert-butyl hydroperoxide and a suitable base generates the title compound, with purification relying on vacuum distillation and careful drying to beat back impurities that could cut shelf life or raise hazards. This process benefits from well-controlled batch reactors, automated temperature monitoring, and regular purity checks, a world away from early bench-scale syntheses that trusted glass thermometers and keen chemical noses. Recent shifts toward greener syntheses look to replace phosgene with carbonylation reactions using less toxic reagents, and such changes show how pressure grows not just from industrial requirements but also from health and environmental advocates.
Chemical Reactions & Modifications
The main draw for tert-butylperoxy-2-ethylhexyl carbonate comes from its power in promoting free radical polymerizations, especially in making polyethylene, polypropylene, and copolymers. The compound pulls apart under heat, sending out tert-butoxyl and peroxy radicals that drive the kick-off of new polymer chains. Scientists have tweaked the base molecule to refine everything from decomposition temperature to solubility profile—modifications like swapping the 2-ethylhexyl group with branched or aromatic variants. These analogs offer lower volatility, tighter control over polymer initiation, or even chances to run reactions under milder, greener conditions. Such changes line up with both the uphill cost of process modifications and the regulatory push for safer chemistries. Laboratory-scale modifications feed pilot-plant innovations, but every tweak faces the same demand for strict safety review before scaling up.
Synonyms & Product Names
Across the chemical industry and research circles, tert-butylperoxy-2-ethylhexyl carbonate appears under several labels. Common synonyms include TBEC, 2-ethylhexyl-tert-butylperoxycarbonate, and tert-butylperoxycarbonate of 2-ethylhexyl alcohol. Trade names may reference the product’s peroxide content or its polymerization role, found under proprietary mixtures from major chemical suppliers. Synonym confusion can run up costs in the lab, so researchers trade stories about shipment delays from minor naming differences—proof that standardized naming and clear CAS registration save headaches as much as they boost reliability.
Safety & Operational Standards
tert-Butylperoxy-2-ethylhexyl carbonate calls for thoughtful handling protocols. Standard PPE includes safety goggles, nitrile gloves, and flame-resistant lab coats. Chemical storage needs rigid temperature control (usually below 30°C) and segregation from reducing agents, acids, and catalytic metals. Industry regulations, anchored in OSHA, REACH, and GHS frameworks, require detailed tracking along each step of production, from raw material entry to finished product storage. Regular training runs keep staff sharp on emergency spill and fire response, which cuts down accident rates. Written safety data sheets drill into specifics on inhalation risk, skin contact protocol, and what to do if the peroxide finds its way outside containment—a scenario that can escalate due to the compound’s ability to stoke combustion or even trigger mild explosive events when mishandled. Site managers share real-world examples of drills and incident reviews, giving new workers practical context for rules that might otherwise read like mere paperwork.
Application Area
tert-Butylperoxy-2-ethylhexyl carbonate shines brightest in polymer chemistry. Polyethylene and polypropylene makers turn to it for reliable free-radical initiation, allowing for robust yield and predictable molecular weight profiles. Tire and elastomer manufacturers lean on its ability to modulate cross-link density, crucial for end-use flexibility and toughness. Resin makers favor it in curing agents, where the moderate decomposition temperature lets them get consistent, bubble-free surfaces with minimal waste. Some specialty uses pop up in the manufacture of foam insulations or certain coatings, where careful temperature ramping steers the reaction toward the desired physical properties. Researchers keep pushing the limits, exploring minor tweaks for biomedical or electronic applications that demand tighter chain control or new stimulus-response profiles.
Research & Development
The last two decades brought a wave of new research into both process safety and performance. Academic labs published on kinetic studies defining precise decomposition profiles, enabling manufacturers to tailor their equipment for less waste and tighter end-point control. Environmental research tracks degradation byproducts in wastewater, nudging manufacturers toward closed-loop systems or advanced etrapment protocols for spent initiators. Digital twins and process simulations now play a large part in pilot plant evaluations, allowing teams to game out outcomes before running costly or dangerous full-scale trials. Industry-academic partnerships focus on replacing legacy synthesis steps with cleaner, less hazardous alternatives and developing formulations with increased stability, extending product shelf life and boosting profits in tough market conditions. Veterans in the field speak about collaborative breakthroughs, where pressure from health and safety regulators steered development teams toward new stabilizers, packaging improvements, and field-testing that gave supervisors the confidence to scale up with fewer incidents.
Toxicity Research
Toxicologists keep a close eye on acute and chronic effects tied to tert-butylperoxy-2-ethylhexyl carbonate exposure. Animal model studies show low acute toxicity by ingestion, but inhalation and skin contact hazards draw sharper concern, prompting close monitoring in plants and labs. Chronic exposure research, especially from mouse and rat models, highlights potential respiratory tract irritation and possible links to mild neurotoxicity, emphasizing the need for airtight ventilation and precise air monitoring in work areas. Environmental toxicologists track breakdown products in aquatic environments, with current research keeping tabs on bioaccumulation and the effect of low-level peroxide residues on local ecosystems. Fact-based data, not rumor or conjecture, form the backbone of the training required to get a signoff for handling this material. Ongoing surveillance, including both human biomonitoring and improved animal studies, may turn up more subtle effects, which underscores the need for continuing investment in safety science and regulatory updates.
Future Prospects
tert-Butylperoxy-2-ethylhexyl carbonate stands ready for new waves of innovation, especially as industrial chemistry leans harder on sustainability and energy savings. The drive for safer, greener production methods will keep stressing greener synthesis, lower-waste routes, and bio-based feedstocks. Product development teams invest in packaging technology that extends shelf life and cuts down on accidental releases. Within the next decade, hybrid approaches using renewable energy for temperature control and digital monitoring to catch early signs of instability could shift how plants manage peroxide initiators. Application research into specialty polymers, high-value elastomers, and electronic materials promises to broaden the reach and market for compounds like this, buoyed by data-sharing across borders and robust regulatory oversight. Over the long haul, the chemical’s role looks set to remain solid, propped up by a blend of safety improvements, growing industrial demand, and ongoing research into safer, more sustainable process designs.
Chemistry Behind the Scenes
Behind many of the things we use every day—plastics, rubbers, coatings—there’s a line of unsung chemical helpers doing important work. Tert-Butylperoxy-2-ethylhexyl carbonate is one of those. It falls under a class of chemicals called organic peroxides. These aren't household names, but their fingerprints show up just about everywhere in modern manufacturing.
This compound works as a radical initiator. In plain language, that means it sets off reactions that turn small molecules, called monomers, into big chains known as polymers. For anyone who's ever wondered what makes tough plastic pipes, flexible phone cases, or weather-resistant window frames, this is it: starting those chemical chains at just the right moment. Getting molecules to link up on command isn’t magic—it’s careful chemistry, and companies rely on initiators like this one to keep products coming out tough and reliable.
Safety and Environmental Responsibility
Working with organic peroxides calls for real attention. Some of them can catch fire if not stored carefully, and a few could pose risks to health or the environment. This brings real-world consequences. Factories handling tert-Butylperoxy-2-ethylhexyl carbonate need trained staff, sturdy containers, and clear safety protocols. These steps aren’t just paperwork; they keep people out of harm’s way and reduce spills that could pollute water or soil.
Standards exist for handling and transporting materials like this. The European Chemicals Agency and regulatory bodies in the U.S., China, and elsewhere set requirements around labelling, storage, and personal protection. Following these means safer workplaces and fewer surprises for communities nearby.
Why It Matters for Manufacturers
Ask anyone running an extrusion line for plastics or planning out an automotive part, and they’ll talk about cost, speed, and reliability. Initiators like tert-Butylperoxy-2-ethylhexyl carbonate fit right into that. Its chemical properties let production lines run at lower temperatures, using less energy than older chemicals. Lower energy means less money spent and a smaller carbon footprint. In places where electricity costs or emissions matter, that’s a pretty convincing sell.
I’ve seen production engineers debate over which initiator type to pick. Some older options break down unpredictably, causing quality issues or even ruined batches. This compound offers more control, especially at temperatures that suit new, high-performance polymers. If your business depends on the difference between scrap and a perfect run, materials like this are part of the solution.
Looking for Safer Alternatives
Not everything about synthetic chemistry is settled. While today's tert-Butylperoxy-2-ethylhexyl carbonate offers clear benefits, researchers keep testing alternatives using greener feedstocks or offering even lower hazard profiles. Brands now ask suppliers for lower-toxicity chemicals and seek recycling systems that keep peroxides out of landfill or water sources.
Collaborations between manufacturers, chemists, and regulators strengthen oversight. For example, some companies automate initiator dosing and switch to smaller containers to limit handling risks. Others invest in fire suppression and regular drills to keep staff ready for emergencies. No one can wave a wand and invent a risk-free chemical, but listening to science and experience drives steady improvements.
The Path Forward
As markets shift and customers demand more sustainable products, the search for better chemical tools won't stop. Tert-Butylperoxy-2-ethylhexyl carbonate holds a valuable place in polymer and plastics production. Its responsible use shows how practical chemistry supports the industries that shape our world, even if its name rarely gets a mention outside the lab.
Getting Down to the Details
Anyone who spends time in a lab knows the importance of being able to read a chemical name and picture its structure. tert-Butylperoxy-2-ethylhexyl carbonate isn’t just a tongue-twister; it’s a mouthful with a real punch in industrial chemistry, especially for polymerization and as an initiator. This specific peroxide stands out because its structure hands over plenty of oxygen to fuel reactive processes.
This chemical’s formula: C13H26O4.
Importance of Structure in Chemical Use
Take a closer look, and the value of tert-butylperoxy-2-ethylhexyl carbonate starts with how its atoms fit together. It has a carbonate group, joins up tert-butyl and 2-ethylhexyl, and puts a peroxide functional group in play. The backbone offers stability, while the weak O–O bond in the peroxide section is ready to break and provide the radical power polymer chemists need. It's not just about blowing up a formula on a chalkboard – this relates directly to curing plastics, resins, or rubbers. When I’ve worked with peroxides, the balance between reactivity and stability means fewer surprises on the bench and more predictable results.
Safety and Handling
Anyone handling peroxides gets the safety training drilled into them quickly. tert-Butylperoxy-2-ethylhexyl carbonate is no exception. The extra oxygen makes for a sensitive material. Heat, friction, or contaminants push it toward violent reactions. Facts tell the story: since peroxides decompose and release energy, facilities store them cold and shielded from sparks. In my own experience, double-checking temperature controls and sticking with only compatible storage matter more than any fancy chemical understanding. Even seasoned hands keep spill kits close. Safety isn’t a glamorous part of chemistry, but it's the one that keeps people safe and businesses running.
Regulatory and Environmental Thoughts
Modern manufacturing can’t ignore environmental responsibility. tert-Butylperoxy-2-ethylhexyl carbonate holds up under many regulations, as large-scale users stay within the thresholds set by REACH in Europe and EPA in the States. It doesn’t take a wonder chemist to see that safe disposal means oxidation under controlled burns, or reaction with reducing agents, not pouring it down the drain. These principles echo across my circles, where compliance is a routine check-in – it’s easier to catch an issue upstream than to explain it to a regulator downstream.
Finding Better Ways
Every new regulation or safety scare gets chemists thinking about alternatives. Greener peroxides, biodegradable polymer options, or tweaks in process temperature and containment have popped up frequently in my career. Firms don’t switch overnight, but the right formula changes — using fewer grams, better containment, or smarter catalysts — bring costs down and risks with them. Tert-butylperoxy-2-ethylhexyl carbonate will keep playing an important role for specialty applications, but the best labs and factories always keep an open mind to safer or more sustainable routes forward.
The Nature of tert-Butylperoxy-2-ethylhexyl Carbonate
tert-Butylperoxy-2-ethylhexyl carbonate brings an air of concern to any workplace handling it. As an organic peroxide, it ranks as a hazardous substance. The chemical draws attention for both its strong reactivity and sensitivity to heat or contamination. This doesn’t just matter for compliance or box-ticking—people working around it can’t afford a slip when talking about peroxides. Every container, cabinet, exhaust duct, and glove carries a piece of the safety equation.
Why Proper Storage Shapes Outcomes
Most accidents with organics like this one do not come out of thin air. A tangle of bad habits—overlooked labels, sloppy segregation, or ignoring temperature warnings—brings real danger. The chemical decomposes rapidly if temperatures rise, unleashing toxic fumes and fire. That’s not an unlikely risk, either: peroxides have a track record of turning routine days into messes none of us want to clean up. After fifteen years working with chemical supply and lab setups, I keep coming back to a simple truth: safe storage shapes outcomes in labs and plants more than any other step.
Temperature Makes All the Difference
Keeping tert-butylperoxy-2-ethylhexyl carbonate cool might sound basic, but it holds life-or-death importance. Organic peroxides often break down above 30°C. If you let this compound hit warm conditions, runaway decomposition can start. Refrigerators—dedicated only to peroxides, away from food or other chemicals—offer one path. Earthquakes, power outages, or careless hands can threaten this fragile balance.
Segregation Prevents Catastrophe
Plenty of disasters start with a small mix-up. A splash of acid or a pinch of reducing metal spells trouble. This compound should never share space with combustibles, acids, bases, or heavy metals. Labels need to be bold and persistent; even a smudge or a worn corner brings confusion and risk. I’ve seen labs invest hundreds in chrome-plated signage and custom racks just to remove guessing games from peroxide handling.
Container Choice and Monitoring Practices
Peroxides like this call for vented, pressure-rated containers. Glass brings shatter risks, while plastics may weaken over time or react. Manufacturers usually specify the right type, and ignoring that advice presses fate. Color coding storage units for organic peroxides cuts confusion during frantic moments. Pairing storage with careful recordkeeping helps even more. A sign-out log sounds bureaucratic, but tracking container movement protects everyone inside the building.
Training and Emergency Planning
People make or break safety. Even the best room, fridge, and label mean little without good training behind them. Regular drills, refreshers on spill kits, and real-world “what if” scenarios keep readiness high. Emergency ventilation and fire suppression gear—never locked, never blocked—provide the last line of defense. Facilities holding tert-butylperoxy-2-ethylhexyl carbonate need up-to-date MSDS sheets and rapid lines to fire services. Sharing lessons from near-misses keeps a workforce sharp and skeptical of shortcuts.
Room for Improvement
Some labs still use decades-old fridges or worn cabinets, betting against bad luck. Investment in smart sensors—triggered by rising heat or open containers—offers a modern solution. Digital tracking and cloud-tied monitoring can warn supervision before things get critical. Funding often stands in the way, but the cost looks small next to the price of fire, injury, and legal action.
In the end, safe storage of tert-butylperoxy-2-ethylhexyl carbonate blends respect for chemistry with day-to-day vigilance. It’s not glory work, but it keeps people safe and production steady—a goal worth a few extra steps and dollars.
Understanding the Risks Lurking in the Lab
Ask anybody who’s spent time around peroxides, and they’ll tell you: respect matters. tert-Butylperoxy-2-ethylhexyl carbonate, a mouthful of a name and a common organic peroxide in the chemical industry, isn’t just another bottle on the shelf. It offers real value in polymerization and related reactions, but there’s a serious safety story behind its usefulness.
Peroxides Turn Up the Heat on Safety
I’ve seen how small spills of organic peroxides can change the whole mood of a laboratory. These chemicals release oxygen radicals with a little push—heat, friction, even stray static. tert-Butylperoxy-2-ethylhexyl carbonate breaks down if you store it warm or knock it around. The heat from this breakdown has been known to start fires in storage—or even, rarely, cause explosions.
The U.S. Occupational Safety and Health Administration (OSHA) keeps a clear eye on organic peroxides. According to safety data from manufacturers, it doesn’t take much heat to move this peroxide into a dangerous zone. Exposed to sunlight or mixed with acids, contamination speeds up the process. That’s not guesswork. Data shows plenty of peroxide-related incidents in factories and research fields.
Skin, Eyes, Lungs—All in the Line of Fire
Tert-Butylperoxy-2-ethylhexyl carbonate won’t do your body any favors on contact. Spills on skin can turn into burns; eyes take a bigger hit. Fumes from the pure stuff put the lungs at risk, often causing coughing or worse in closed rooms. Even the more diluted product smells sharp, and that’s the body warning you to back off.
Safety guidelines recommend working with gloves, goggles, and a decent fume hood. In more than one plant, I’ve watched workers swap out regular gloves for sturdier nitrile ones after noticing the breakdown of their old ones. That’s learning the hard way—but it only takes a single slip-up for that lesson to stick.
Fire Doesn’t Wait for Accidents
In regular work settings, fires linked to peroxides often start from somebody tossing scrap into a trash bin. Adding tert-Butylperoxy-2-ethylhexyl carbonate-soaked material to the weekly trash run creates a risky pile—heat builds as things decompose, and then things burst into flames. I’ve heard more than a few warehouse stories about ruined mornings for this reason alone.
Red Flags and Solutions That Actually Work
People handling peroxides owe their safety to clear labels, regular training, and storage away from sunlight and any sources of sparks. Temperatures need to stay cool—think below 30°C, often using fridges meant only for chemicals. Routine checks, spill drills, and strong ventilation mean fewer accidents.
Suppliers have found safer stabilizers, but even improved versions don’t take away all the risks. Substitution sometimes solves the problem, if another initiator does the job with less heat hazard.
Respecting the chain of custody for waste disposal matters more than people realize. Mixing peroxide-containing materials with general waste never ends well. Professional waste services keep a tight lid on dangerous by-products and keep the rest of us out of the headlines.
Safer Science Starts with Practical Care
Tert-Butylperoxy-2-ethylhexyl carbonate can push industry and research ahead, but sloppy habits turn it from a tool to a hazard. Knuckle-down safety routines, up-to-date training, and real respect for the material keep teams safe. That makes the difference between routine work and a story nobody wants to tell.
Why the Shelf Life of This Peroxide Matters
The clock starts ticking from the day tert-Butylperoxy-2-ethylhexyl carbonate leaves the production line. Like a lot of peroxides, you cannot ignore the calendar with this chemical. It plays a key role in polymerization, cross-linking, and curing in plastics and rubbers. I’ve seen big production batches go wrong because folks assumed those “best by” dates on chemicals didn’t make a difference. They do. Tert-Butylperoxy-2-ethylhexyl carbonate won’t catch fire in the bottle, but as the months roll by, its strength drops. You pay for predictable results, so using a degraded product means slow cures, incomplete reactions, and wasted time.
The Science Behind a Reduced Shelf Life
Everything starts with its chemical structure. This compound contains a peroxide bond—essentially, an oxygen-oxygen bridge. That bond is why it starts reacting so easily, but it’s also why it won’t last forever. Heat, light, and oxygen in the air push that bond to split before you ever mix it into a resin or plastic. Once those bonds start breaking, you’re looking at lowered activity and potential hazards.
The manufacturer will usually recommend a shelf life of about six months to one year if stored cold and in original packaging, away from sunlight. Keep it at room temperature or forget temperature logs, and the shelf life drops off even faster. I once checked a warehouse where a batch sat on a hot shelf in direct sun all summer—the storage manager found white residue and a sulfur odor. By then, you can’t trust the product for any real work. In fact, you may need to call in a waste disposal crew for any leftovers.
Protecting Your Peroxide Investment
Strict storage is not just a cautionary note—it saves money and prevents injuries. The bottle should stay in a dedicated flammables fridge, temperature set between 2°C and 8°C, well away from acids, bases, and anything flammable. Containers should seal tightly after each use. Wearing goggles and gloves while handling this stuff sounds basic, but I watched a new lab tech slap on just a dust mask, thinking it’d be good enough. Later, peroxide vapor escaping from a poorly closed bottle set off an alarm.
Safety data sheets (SDS) from the top suppliers spell out that even a small amount left exposed to air will lose potency. They back it up using accelerated aging studies, tracking how fast the active oxygen content drops off. I go right back to the original labeling instead of guessing. If that bottle got opened nine months ago, and storage records show it spent time at twenty degrees, I schedule a fresh order rather than risking a process failure or unexpected pressure build-up in the lab waste drum.
Keeping Control on the Factory Floor
For operations that rely on tert-Butylperoxy-2-ethylhexyl carbonate, shelf life ties directly to quality control. Experienced technicians rotate stock, mark the open dates, and use up older bottles first. Some companies set automated reminders to pull and test lots nearing expiration, checking the active oxygen level with iodometric titration. Managers push for regular inventory checks because people make mistakes, and it’s easy to forget a carton behind new shipments. With proper routines, most chemical incidents can be avoided before they start.
Instead of gambling on uncertain potency, smart teams trust the manufacturer’s recommendations, run pilot tests, and invest in solid storage. They treat these shelf life limits as hard deadlines, not suggestions. That approach keeps both final products and the people making them a lot safer.