Exploring Tert-Butyl Peroxypivalate: From Lab Bench to Industry Floor
Historical Development
Back in the middle of the twentieth century, folks in chemical labs started hunting for organic peroxides that could act as radical initiators in polymer production. Tert-butyl peroxypivalate showed up on their list after researchers discovered its knack for helping certain plastic and resin reactions along at relatively mild temperatures. This was significant during the early days of polymer science, largely because reliable initiators often made or broke the process—mess up your catalyst, end up with junk in the reactor. Over time, chemists figured out its sweet spot for temperature and how to avoid the riskier behaviors associated with some other peroxides. Companies that wanted to expand plastic manufacturing praised this compound for the control and efficiency it offered compared to options floating around at the time.
Product Overview
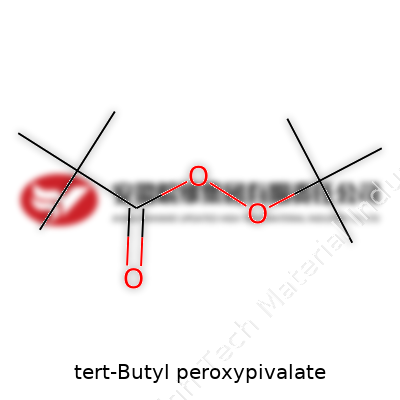
Tert-butyl peroxypivalate is an organic peroxide that serves as an initiator, mostly in polymer chemistry. Manufacturers package this liquid or stabilized solution and ship it under constraints that keep temperatures low. The chemical structure—marked by a tert-butyl group connected by a peroxide bond to a bulky pivalate section—delivers reliability in starting free-radical reactions required in plastic factories. Bulk suppliers label it by its many trade names, but the main thread is the combination of tert-butyl and peracetic acid units. Factories use it because the decomposition profile fits snugly into processes for making PVC and other specialty resins.
Physical & Chemical Properties
Tert-butyl peroxypivalate comes as a colorless to pale yellow liquid. Its molecular weight, around 202 grams per mole, keeps things balanced between volatility and function. The compound’s boiling point sits far enough below room temperature that storage in a deep freezer or with ice packs becomes a must. It reacts briskly with organic materials when heated, making safety a top concern. I’ve seen plant engineers fret about storage conditions, especially when summer temperatures drive up ambient heat. With a low flash point and an ability to decompose into volatile products, this peroxide demands careful attention to containment and transport.
Technical Specifications & Labeling
Commercial shipments describe tert-butyl peroxypivalate in concentrations ranging around 75% by weight, typically cut with phthalates or similar solvents to tone down its energetic side. Labels spell out the temperature limits, usually no warmer than 30°C for transport and much cooler for storage, with danger symbols prominent for organic peroxides. Product data sheets list details about percentage purity, recommended handling equipment, maximum shelf life (if the product is handled correctly), and compatibility with stabilizers or inhibitors. The specifics on certificates of analysis—peroxide value, water content, acidity—help quality control labs confirm batches still fall within performance windows.
Preparation Method
Manufacturers synthesize tert-butyl peroxypivalate by reacting tert-butyl hydroperoxide with pivaloyl chloride, usually in the presence of a weak base. The two starting materials combine, releasing hydrochloric acid gas, so production setups anchor around excellent ventilation and robust scrubbing systems. This method churns out a product ready for dilution, purification, and stabilizer addition. The batch gets dried, chilled, and sealed away to keep stray heat and ignition sources far out of reach. Companies might adjust reaction temperature, stirring speed, or base concentration to tweak yields and purity. Technicians test each run’s active oxygen content to track consistency across lots.
Chemical Reactions & Modifications
In a plant setting, tert-butyl peroxypivalate stands out for its clean, predictable radical formation when exposed to moderate heat. It can initiate chain-growth reactions—such as polymerizing vinyl chloride or acrylates—at temperatures below 60°C, which benefits plant energy bills and keeps unwanted side reactions to a minimum. Certain processes tinker with the molecule by swapping out the solvent carrier, diluting the stock, or pairing it with co-initiators that modify polymer architecture or molecular weight. Labs studying its reactivity learn to appreciate how additives (like stabilizers) or trace metals shift decomposition rates, ultimately affecting polymer characteristics.
Synonyms & Product Names
Walk into any chemical warehouse and you’ll find tert-butyl peroxypivalate under a handful of synonyms. Some call it TBPP, others might reference pivalic acid, peroxydi-tert-butyl ester, or peroxypivalic acid tert-butyl ester. Trade names—whether from AkzoNobel or Arkema—camouflage the substance behind proprietary branding, but the function stays the same. Distributors require everyone on the handling chain to match synonyms to the correct safety guidelines to prevent mix-ups, since organic peroxides as a family contain many close cousins but with very different hazards.
Safety & Operational Standards
Anyone who works with tert-butyl peroxypivalate talks about safety until they’re blue in the face. Plant managers drill new hires on the need for goggles, gloves, flame-resistant lab coats, and proper ventilation. This material goes volatile with very little provocation—heat, friction, shock, or contamination can trigger runaway reactions. Storage facilities need non-sparking tools, temperature alarms, and backup cooling systems. Emergency plans call for containment, neutralization, and coordinated evacuation if spills happen. Regulatory agencies such as OSHA and the European Chemicals Agency pay close attention to safety documentation. Training refreshers, risk assessments, and meticulous housekeeping routines help keep accidents to a minimum and make sure everyone leaves as healthy as they arrived.
Application Area
Resin manufacturers value tert-butyl peroxypivalate for the gentle hand it offers in starting vinyl chloride and acrylate polymerization. It’s a go-to initiator in production of PVC pipes, window frames, and certain plastics used in automotive and construction. Blending it into polymerization tanks allows for tight control of reaction timing, product strength, and color. I’ve watched technical teams swap initiators out during process optimization trials, trying to maximize yield and minimize unwanted branching or crosslinking. Some specialty adhesives and coatings also use this compound for its ability to kick polymers off without scorching the product or fouling reactor walls.
Research & Development
Academic labs and industrial R&D outfits continue picking apart the details of peroxide chemistry to design safer or more efficient materials. Chemists tinker with molecular structure, hoping to lower decomposition temperatures or reduce environmental impact. Technicians trial new stabilizers or test compatibility with greener solvents. Patents pop up for process tweaks that stretch product shelf life or curb hazardous waste. Some research projects focus on recycling initiator byproducts; others try to replace older catalysts, cutting down on byproducts altogether. Competition between global chemical suppliers sparks new formulations, giving end users more tools to push polymer performance further.
Toxicity Research
Researchers agree that tert-butyl peroxypivalate poses hazards on skin contact, inhalation, or accidental mixing with incompatible chemicals. Toxicology studies show that even short exposure can irritate eyes and respiratory passages, and chronic exposure leads to bigger health risks. Acute effects include headaches, dizziness, or worse—detailed medical case studies follow factory mishaps, reminding management to keep air monitors and fume hoods in top shape. Disposal crews must treat all residues as hazardous waste and follow strict protocols. Regulatory filings in the US and Europe list the substance on controlled chemical inventories, reinforcing rules around personal protective equipment and workplace exposure limits.
Future Prospects
As consumer demand for plastics grows, chemical companies keep searching for safer, cheaper, and more precise initiators. Tert-butyl peroxypivalate, with its track record in global production, isn’t losing relevance soon. Ongoing research pushes for initiators that lower the risk profile, especially for workers in small or under-resourced plants. Green chemistry programs encourage a move toward renewable raw materials, leaner synthetic routes, and biodegradable byproducts. Partnerships between industry and academia invite fresh ideas—whether switching out solvent carriers, tweaking the core molecule, or monitoring batch consistency with big data. The next wave of innovation might reduce environmental impacts or deliver even greater control over product qualities.
Producing Plastics: The Main Role
Tert-Butyl peroxypivalate plays its biggest part in making plastics. Most people never see it up close, but it sets off the chemical reaction that turns raw monomers into polymer chains. I remember touring a small plastics facility on the edge of town and watching giant vats of resin bubble away. The plant operator explained that without specific chemicals—like tert-butyl peroxypivalate—this process doesn’t get off the ground. It’s the spark that moves things along, ensuring the final product comes out with the right strength and flexibility.
Acrylics, which show up in paints, adhesives, and even contact lenses, rely on a steady reaction pace. Tert-butyl peroxypivalate gets chosen for these jobs because it kicks off reactions at lower temperatures. That means less wasted energy and more stable quality control on the factory floor.
Supporting the Paint and Adhesive Industry
Anyone who’s ever rolled a glossy coat of paint on a living room wall or pressed together laminated countertops has encountered products shaped by this chemical, even if they never see the bottle. Paints need to stay smooth, level, and bright under tough conditions. The same goes for adhesives—those hidden glues holding shoes together, or the sticky stuff in plywood. This chemical helps companies dial in just the right properties by managing the polymerization step with accuracy.
I’ve chatted with workers at a flooring manufacturer. They said switching to alternative initiators sometimes caused bubbles or inconsistent texture in their product. With tert-butyl peroxypivalate, they kept failures low and quality up, saving hassle and reducing waste. The fact that it works at lower temperatures and doesn’t break down as fast helps a busy production line run without as many hiccups or delays.
Risks and Worker Safety
Working with any peroxide brings real risks, and tert-butyl peroxypivalate isn’t different. It breaks down quickly if not handled right, and that can cause dangerous fires or releases. Factory floors train crews to keep storage areas cool and monitor for leaks. I’ve spoken with older chemical handlers who recall accidents from improper storage in the past. Many factories now use automated dispensing and strict temperature monitoring to keep things safe.
The material’s risks push producers and buyers to evaluate handling protocols every year. New sensors and more accurate temperature controls cut down on unforeseen problems. Although the chemical itself never leaves the plant, making these improvements protects workers and communities nearby.
Looking for Better Options
Some manufacturers hope to phase in safer alternatives with similar efficiency and less volatility, but established supply chains and cost pressures keep tert-butyl peroxypivalate in widespread use. There’s movement—especially in Europe—toward processes that use greener chemistry. Some new labs experiment with bio-based initiators or try to cut down on hazardous by-products.
Government agencies in many countries require documented training and emergency plans before use. They publish guidelines and push companies to report safety incidents. Over the years, this oversight has raised the bar for chemical manufacturing, but real-world results depend on companies investing in the right equipment and ongoing learning for the workforce.
Weighing Benefits Against Risks
Tert-butyl peroxypivalate may not make headlines often, but it shapes parts of modern life most of us touch every day. Progress means looking for ways to do more with less risk, and that focus on safety and innovation almost always starts at the factory floor. Regular reviews and investments can keep benefits high—whether making car parts or safer kitchen counters—without putting health or the environment on the line.
Getting the Formula Right
Every chemist who’s spent time in a teaching lab knows the feeling—one missed atom on a molecular drawing, and the instructor pounces. It only takes a simple slip for a substance to become something completely different. With tert-Butyl peroxypivalate, also known as TBPP, the chemical formula isn’t just some abstract string for textbooks. It affects everything from how it’s handled in industrial plants to how it’s listed as a hazardous material. The formula reads: C9H18O4.
How the Structure Connects to Safety
Looking at the formula, C9H18O4, you see a total of nine carbons, eighteen hydrogens, and four oxygens. TBPP belongs to organic peroxides—compounds famous for their ability to decompose and release oxygen. Chemists learn to respect these peroxides, because any error in understanding their make-up could send processes off the rails. TBPP is sometimes used as a polymerization initiator. This just means it helps start reactions that link smaller molecules together to make plastics. But its peroxide group is sensitive—if the chemical breaks down outside of controlled settings, it can lead to fires or explosions.
Why This Formula Makes a Difference
The details tucked inside C9H18O4 end up guiding real-life decisions. Companies rely on laboratory data to find out how stable the substance is, how much heat it can handle before breaking down, and what kind of containers may be trusted for storage. If a manufacturer were to misunderstand the formula, the rest of the safety net could unravel. I spent a summer in a polymer lab where the difference between methyl ethyl ketone peroxide and benzoyl peroxide was drilled into our heads. One slip-up, and the risk wasn’t just lost product, but real danger for every technician in the room.
Supporting Evidence for Accuracy
The need for concrete chemical formulas isn’t based on tradition, it ties directly to safety protocols and regulatory compliance. The United States Occupational Safety and Health Administration (OSHA) lists tert-Butyl peroxypivalate under their extremely hazardous substances for a reason. The Environmental Protection Agency (EPA) monitors its usage due to its flammable, reactive nature. Material Safety Data Sheets (MSDS) reflect the C9H18O4 formula and include tailored handling instructions—everything from temperature controls to proper venting. Getting this detail right can affect how insurance companies underwrite chemical storage or how emergency teams respond in a spill.
Room for Improvement
Colleges and training programs should go beyond rote memorization by giving more hands-on experience with compounds like TBPP. If students prepare tert-Butyl peroxypivalate in small, tightly controlled batches, they see firsthand why every atom counts. Commercial labs can extend this care by double-checking labels and running regular audits—it’s never a wasted effort. Regulatory agencies deserve support expanding education and offering easy-to-read guidance, so not just experts keep up, but new hires and non-specialists stay safe around active chemicals.
Looking Forward
Honest discussions about chemicals shouldn’t stay locked away in textbooks. Real people use these substances daily to build things, clean things, and power innovation. Getting the formula right brings practical benefits—accurate safety protocols, easier compliance, and fewer accidents. For tert-Butyl peroxypivalate, knowing C9H18O4 by heart is just the start. Beyond memorization, the formula serves as a critical checkpoint between science, safety, and progress.
Understanding the Risks of Tert-Butyl Peroxypivalate
Tert-Butyl peroxypivalate, used widely as an initiator in polymer production, comes with some unique hazards. Folks working with it see pretty quickly that this isn’t just any chemical. It breaks down easily and can release energy in ways that catch people off guard. More than a few fires or explosions have started with people thinking they could cut corners. Handling this compound involves more than reading a safety data sheet once and locking it away on a shelf.
The Importance of Temperature Control
Heat makes things go wrong with tert-Butyl peroxypivalate. Once the temperature starts climbing, decomposition reactions pick up speed, and there’s a real risk of fire or explosion. Temperatures should stay under +20°C. In my time consulting for a plastics plant, lab techs learned by experience to check the storeroom thermometer several times a day in summer, and by night as well. Dry ice or cold packs in secondary containment often gave just the buffer needed when the regular fridge failed, especially during heatwaves.
Choosing the Right Storage Location
Regular stockrooms or open shelving sets up trouble. Tert-Butyl peroxypivalate goes in a separate flammables cabinet, ideally a dedicated explosion-proof refrigerator. Vented cabinets with automatic fire suppression cost more, but stories from other sites convinced us: repair bills and insurance claims look cheap next to lost lives. Storage away from sunlight and heat sources protects everyone. Even strong overhead lights can heat non-insulated cabinets, leading to dangerous pressure buildup in bottles.
Container Quality and Labeling
Containers matter as much as the room itself. Only containers made of materials recommended by the supplier keep chemicals contained and degradation slow. Metal drums, glass bottles, and certain plastics suit different quantities, but leaks or cracked caps spell disaster. Spending a few extra bucks on certified, tested closures paid off, especially with regulators checking records twice a year. Clear labels don't just satisfy inspectors—they save headaches when people rotate chemicals or clean up old stock.
Avoiding Cross-Contamination and Incompatibility
Never place tert-Butyl peroxypivalate alongside acids, bases, or oxidizable organic substances. A spill, even a small one, brought together with the wrong chemical produces results that can scare even the seasoned worker. I watched someone use a common spatula to wipe up a drip once, only to trigger smoke and a very close call. Separate tools, cleaning supplies, and secondary trays for spills or drips keep people safe.
Emergency Planning and Routine Inspections
No amount of preparation replaces routine walk-throughs and drills. Clearly marked spill kits, updated training, and paperwork that gets checked weekly help small problems show themselves before they become big disasters. Working with a local fire department or hazardous materials team paid dividends not just in incident response but in peace of mind during audits.
Finding Lasting Solutions
Addressing the root causes—old buildings, failing cooling systems, poor layout—demands more than checklists. Investment in modern safety upgrades, real conversations with workers, and constant vigilance turn a risky material into just another part of the job. People deserve to go home every day in one piece.
What’s at Stake?
tert-Butyl peroxypivalate doesn’t mess around. It acts as a powerful organic peroxide, making it a favorite in plastics and resins for initiating polymerization. But nothing about this stuff feels ordinary. Its reactivity and risk call for a careful approach, both for the person handling it and the facility itself.
Understanding the Dangers
One whiff of tert-Butyl peroxypivalate burns the memory into your nose. Vapors can irritate the eyes, skin, and lungs in seconds, and if you spill it or heat it up, you’re facing a fire hazard few want to deal with. At the plant, nobody forgets how much attention a drum of this chemical demands—because stories spread fast after injuries or explosions. The National Fire Protection Association marks these peroxides with a bold "Reactivity" warning, and for good reason.
Handling Starts with Preparation
You never catch an experienced technician pouring or measuring tert-Butyl peroxypivalate without protection. Thick chemical splash goggles, nitrile gloves up the wrist, and an apron that won’t give out if something splatters—this is standard issue. It always helps to check the safety datasheet before getting started. Fresh air isn’t optional. Proper fume hoods or local extraction fans must run full blast to keep fumes out of your lungs. Once, I learned the hard way what a few missed drops on bare skin can do: nasty redness in minutes and a story I retell any time a new hire skips their gloves.
Storage Means Respect
This chemical always goes straight into a temperature-controlled fridge or cooler room—never on open shelves or near a heat source. Warm storage turns this compound into a time bomb. Tanks and containers need plenty of space around them to avoid bumps and pressure build-ups. At the lab, a locked cabinet marked with both hazard and “Organic Peroxide” warnings keeps wandering hands away. Anyone in the facility knows to treat leaks or even broken vials as emergencies.
Training and Culture Save Lives
A good safety culture changes everything. New staff go through emergency drills with mock spills before touching the real chemical. After one close call, our supervisor made sure no one could open a new container until they could demonstrate every safety step. This culture of double-checking each other—never skipping steps, never rushing—turned out to be the most effective precaution.
What If Something Goes Wrong?
Emergency eye showers and safety showers stay free and clear—not blocked by boxes or notes. Spill kits suited for peroxide cleanup stand nearby, not just generic absorbent. Quick action reduces harm. If a fire starts (and it can, fast), only specialized extinguishers rated for chemical fires work. Training tells us not to play hero. Call the fire brigade and evacuate.
Building a Safer Workplace
It makes sense to run regular refresher training tailored to peroxides like tert-Butyl peroxypivalate. Facilities with fewer incidents usually audit their chemical stocks every month and survey staff on what could make storage and handling safer. Management that actually invests in safer equipment, containers that withstand pressure, and monitoring alarms help keep the next shift from turning tragic headlines.
Handling tert-Butyl peroxypivalate isn’t about ticking boxes—it’s personal. Daily choices and a commitment to looking out for one another set apart the operations that keep moving forward from those grinding to a halt.
What Is Tert-Butyl Peroxypivalate?
Tert-butyl peroxypivalate shows up in places you'd mostly expect if you've ever set foot in a chemical lab or worked in plastics. Folks who use it know it as an organic peroxide catalyst, giving a helpful boost to certain reactions, especially in making polymers and resins. Sounds useful, right? But that convenience comes with baggage.
Why Safety Matters with Organic Peroxides
Experience with organic peroxides tells me: respect the risks. These chemicals love to decompose, which means heat and sunlight aren't just unwelcome guests—they’re downright dangerous. Tert-butyl peroxypivalate falls squarely in this category. Its structure allows it to split into pieces pretty easily. That splitting releases energy. If you’re talking about small amounts, you may get away with some fumes and heat. But if you’ve got a larger batch and the conditions aren’t under control, you’re flirting with an outright explosion.
Hazards in the Real World
Even a quick glance at incident reports paints a picture: mishandled organic peroxides have led to injuries, property loss, and sometimes worse. I remember reading about a warehouse fire that turned out far worse because chemicals like this weren’t properly stored. Tert-butyl peroxypivalate doesn’t need a spark to join the party; temperatures over 20°C (68°F) already move things into dangerous territory. It releases oxygen and other gasses, which can make flames worse and turn a small fire into something you'd see on the evening news.
Explosiveness: More Than Just a Word
Tert-butyl peroxypivalate earns a spot on safety data sheets as both flammable and explosive. The explosion risk isn’t just lab myth—it’s come up in practice. I spoke to someone in industry who said that even experienced hands need a healthy dose of caution. Even gently shaking some older containers can lead to disaster. No surprise—you’re dealing with a chemical that breaks down with a bang, not with a whimper.
How To Keep Disaster at Bay
The good news is, with knowledge and planning, you can keep things safe. Chemical companies use containers that vent safely, and cold rooms help keep the temperature low. Extra training goes a long way. Anyone working with tert-butyl peroxypivalate needs to prepare for accidents before they happen. That means safety goggles, gloves, flame-proof coats, and clear rules about where and how to store the chemical. I’ve seen people forget the label and pay the price. So many accidents start with someone saying, “It looked safe enough.”
Beyond proper storage, there’s a need for communication. Emergency teams must know exactly what’s on hand. Material safety data sheets don’t just stay in a drawer—they need to be on hand, readable, and understood. Regular drills save lives. Open conversations about near-misses help not just one company, but the whole industry learn and adapt.
In summary, tert-butyl peroxypivalate isn’t something to fear blindly, but it earns every word of caution printed on its label. Staying alert, respecting its power, and talking openly about risks—that’s how you keep workplaces safe and quiet instead of making headlines for all the wrong reasons.