Understanding tert-Butyl Peroxymaleate: An In-Depth Commentary
Historical Development
Chemistry rarely spotlights molecules like tert-Butyl peroxymaleate, but it should. The story of peroxides traces back to the foundation of industrial organic synthesis. Researchers in the twentieth century began investigating peroxy esters for their ability to generate free radicals, which serve as the backbone for polymerization technology. In the 1960s, synthetic routes matured, and specialties like tert-Butyl peroxymaleate started popping up, offering new avenues for chemists needing precise control over reactivity. As new industrial applications drove experimentation, this molecule carved out a space that ferried science from laboratory curiosity to essential process tool.
Product Overview
tert-Butyl peroxymaleate isn’t a household name, but anybody working in polymer chemistry or controlled oxidation will recognize its role. Here’s a substance that brings both oxidizing punch and a structure ready for modification, blending the properties of an organic peroxide with maleate’s flexible backbone. Peroxide initiators, including this one, fuel polymerization reactions, facilitating everything from plastics to specialty rubbers. Manufacturers value this product for its trigger-happy initiation under controlled temperatures and its compatibility with diverse monomers. These factors push demand, especially in regions with vibrant chemical industries.
Physical and Chemical Properties
This molecule shows up as a pale yellow liquid or sometimes as a crystalline solid depending on storage conditions and concentration. With its oxygen-oxygen bond, it carries that distinctive sharp smell common to many peroxides, warning workers to handle it with respect. Melting points usually hover between 20 and 35 °C, but the real story comes from its active oxygen content and how rapidly it decomposes above 50 °C. Chemists working with it watch out for the explosive risks that come with handling strong peroxides; tert-Butyl peroxymaleate lives up to the expectation. Solubility sits in most common organic solvents, which simplifies handling in complex synthesis procedures.
Technical Specifications and Labeling
Before cracking open a drum, a chemist checks labeling: manufacturer, purity (often above 98%), active oxygen content (around 7-8%), and impurity profile. You’ll see storage instructions that plainly spell out the risks: keep cool, avoid sunlight, never allow contamination with strong acids or reducing agents. Technical data sheets describe key decomposition temperature and detailed instructions for use, as safety and performance both depend on following these numbers to the letter. DOT labels mark it as an oxidizer and demand proper paperwork from shipping to warehouse to lab.
Preparation Method
Making tert-Butyl peroxymaleate calls for skill and caution. To synthesize it, manufacturers usually react tert-butyl hydroperoxide with maleic anhydride under carefully controlled conditions. Catalysts—often acids—help push the reaction, but strict temperature control is essential to prevent runaway decomposition. Every stage gets monitored closely: water, air, or trace metals can spark unintended reactions, so glassware and solvents need to stay rigorously clean. This process rewards experience and careful attention more than any computer simulation or textbook recipe.
Chemical Reactions and Modifications
In reactions, tert-Butyl peroxymaleate unlocks chains of radicals, driving polymerizations or serving as a stepping stone for further modification. It holds up well in both solution and bulk reactions, making it flexible for industrial setups. Chemists often tweak its structure, replacing substituents on the maleate ring to change decomposition rates or temperature profiles. In complex syntheses, one mistake—like adding incompatible functional groups—can spark violent decomposition, so every modification undergoes rigorous testing and validation on small scales before moving into production.
Synonyms and Product Names
Name changes create confusion in the chemical world. Over decades, tert-Butyl peroxymaleate has appeared under different names—tert-Butyl peroxymaleate, TBPM, and sometimes t-Butyl maleate peroxide. Each label carries the same risk profile, so buyers and researchers double-check CAS numbers and supplier certificates to confirm the product. Proper naming ensures continuity in safety documentation and avoids costly missteps in purchasing and logistics.
Safety and Operational Standards
The harsh lesson with organic peroxides: complacency leads to accidents. Workers need gloves, goggles, and fume hoods—no exceptions. Storage must never approach the decomposition point, and every batch gets stored in explosion-proof containers, away from sources of ignition. Patchy training or slipshod handling courts disaster, and regulatory frameworks like OSHA and REACH have responded with strict guidelines. Chemists routinely run small-scale thermal stability tests to catch any sign of instability before full-scale use. Anyone skipping safety data sheet reviews or handling protocols risks not only product loss but injuries that scar lives and reputations.
Application Area
Industries driving high-value polymer production often reach for tert-Butyl peroxymaleate. The commercial push for better plastics, high-resilience rubbers, and customized copolymers all trace back to initiators like this, which give engineers finer control over molecular architecture. In adhesives, coatings, and even detergent builders, it finds uses where reliable initiation of controlled oxidation makes the difference between average and premium products. Some advanced synthetic labs experiment with its oxidizing potential to tweak pharmaceuticals or develop new agrochemical intermediates, relying on its known decomposability for select cases where milder oxidants fall short.
Research and Development
Laboratories worldwide continue to probe the nuances of this molecule. Teams work on finding more stable or less hazardous analogues, tuning decomposition temperatures, and extending shelf life. As green chemistry continues to rise in prominence, the hunt has begun for preparation routes that avoid heavy metal catalysts or produce less toxic byproducts. Publications from both academia and industrial research groups share new applications or improvements in handling, fueling further innovation. Cross-disciplinary collaboration—especially between polymer chemistry and process engineering—plays a huge role in advancing the field.
Toxicity Research
Working with potent oxidizers brings risks, and scientific literature doesn’t shy away from the frank discussion about toxicological impacts. Studies show tert-Butyl peroxymaleate’s harmful potential on skin, its volatility, and its impact on respiration if inhaled. Chronic exposure or improper disposal creates enough hazard to justify tight workplace exposure limits and robust ventilation systems. These concerns press regulatory bodies to fund deeper investigation into long-term environmental toxicity, potential byproducts, and safer handling protocols. Companies have incentive, too: lawsuits and regulatory fines sting, but reputational loss bites deeper.
Future Prospects
The future points toward safer, greener chemistry but won’t leave molecules like tert-Butyl peroxymaleate behind. As manufacturing standards tighten and society demands environmentally sound processes, the pressure builds for better safety monitoring, lower-waste synthesis, and thorough life-cycle analysis. Emerging research into controlled polymerization, precision catalysis, and biodegradable materials opens fresh doors for exploration. New generations of chemists, trained in both fundamentals and modern environmental stewardship, will continue to redefine how this compound fits into a sustainable industrial chemical landscape.
The Role of tert-Butyl Peroxymaleate in Industry
Walking through a chemical manufacturing plant, I once noticed a set of barrels marked with long, tongue-twister chemical names. Among them: tert-Butyl peroxymaleate. This compound grabs a quiet but important place in industrial chemistry. Companies often keep it in their toolkit for managing tough polymerizations. When manufacturers want to push materials into new shapes and forms, they turn to specific chemicals that kickstart the process. Tert-Butyl peroxymaleate lands in this spotlight for its role as an initiator.
Polymer Production: Shaping Plastics and Resins
Over the years, I’ve heard a lot about complex molecules driving big changes in daily life. Tert-Butyl peroxymaleate operates as a free-radical initiator. That means it sets off chain reactions in the production of plastics, synthetic rubbers, and certain resins. Think about the sturdy water bottles or the smooth dashboard of a car—these come from reactions that rely on timing, temperature, and strong initiators. Without the right spark, the chain reaction stalls, and nothing forms. Factories choose tert-Butyl peroxymaleate in cases requiring controlled and reliable activation. It gives manufacturers an edge, producing materials that don’t crack or break as quickly.
Why This Chemical Stands Out
Their teams need stability, but they also need performance during the heat of reaction. Tert-Butyl peroxymaleate offers both. Many peroxides break down when stored for weeks or exposed to warmth. This compound holds its own longer than some alternatives, so workers don’t worry about ruined batches. Even a small amount can drive a reaction that produces tons of finished polymer product. This reliability matters in industries fighting to keep costs down without cutting corners on safety or quality.
Handling Safety Concerns
I’ve visited enough plants to know no one treats peroxide compounds lightly. Accidents do happen if people get complacent. Tert-Butyl peroxymaleate can decompose vigorously, releasing heat and potentially dangerous fumes. Companies must follow firm safety protocols and train staff to respect storage and handling guidelines. This keeps workers safe, keeps the environment protected, and avoids damaging episodes that shut down production lines for weeks.
Looking at Environmental and Workplace Risk
Environmental questions come up every time chemicals connect to industrial-scale production. Tert-Butyl peroxymaleate itself isn’t something you want spilled in a river. Disposing of leftover material requires planning and adherence to government regulations. Plants investing in better training and leak prevention strategies see fewer mishaps. By pushing suppliers to use safer containment or switch to greener alternatives, companies also support a future where polymer manufacturing won’t mean sacrificing safety or clean air.
Finding Practical Alternatives
Some industries look for alternatives to tert-Butyl peroxymaleate, hoping to reduce workplace risk or lessen environmental impact. Researchers experiment with less hazardous initiators or design closed systems that reuse chemicals again and again. This shift slowly changes how everyday things get produced, from flexible hoses to medical tubing. Even so, for requirements demanding tough, predictable reactions, tert-Butyl peroxymaleate still holds a firm spot in the chemical playbook.
Making Responsible Choices in Modern Manufacturing
People outside factories rarely think about what happens behind the scenes. Yet, every time you touch a synthetic product, there’s a trail of decisions leading back to compounds like tert-Butyl peroxymaleate. By selecting safe, effective solutions and tracking the science, chemical producers continue the push toward safer, cleaner, and more reliable manufacturing for daily goods that people rely on.
The Risks Outweigh the Curiosity
tert-Butyl peroxymaleate comes with a reputation in the lab for being unpredictable. Years ago, I worked alongside folks in a university setting who handled organic peroxides like this one—each time, the room seemed more tense. Their bright white powder looks harmless, but it packs a punch that can surprise even veteran chemists. It isn't just about knowing a few rules; it’s about respecting a material that doesn’t forgive mistakes.
Get Real About Protective Gear
Personal protective equipment becomes a non-negotiable. Lab coats don't always block splashes, so a chemical-resistant apron gives real peace of mind. Goggles are standard, but a full face shield offers more coverage when splashing or explosions could send material across the room. Gloves made of nitrile or neoprene help, since they hold up better than your basic latex pair. Some folks pull on two pairs just to be sure—this extra layer can save a trip to the ER if something goes wrong.
Work in a Space Designed for Trouble
A fume hood isn’t just for ventilation. It draws fumes, but it also provides a small barrier if something ignites or explodes. In my own experience, anything with “peroxide” in the name belongs nowhere near open flames, static electricity, or even sunlight. tert-Butyl peroxymaleate breaks down fast and can spark a fire without much warning. Ground your glassware. Every metal stirring tool or scale should be checked for stray sparks. Saying “no shortcuts” in storage and disposal matters, too—locked cabinets, away from all heat sources, keep accidents in check.
Handling Storage Like a Pro
Don't ever store peroxides in bulk unless it’s essential. Small containers limit the disaster if they go off. Avoid using glass stoppers that freeze shut, forcing you to twist and risk breakage. Label every container as if the next person opening it has never seen it before, and always date containers—peroxides degrade, and old stock won’t give a warning before turning dangerous. To prevent spills from spreading, line shelves and keep chemical absorbents within reach.
Leaks and Spills: Own Your Response
If you spill, back away, inform others, and grab spill pillows or vermiculite—not paper towels. Wipe-ups cause friction and heat, which set off peroxides. Immediately minimize the area, and always approach a spill kit as your first tool. Call in environmental services before you think you need them; peroxides wait for nobody. I learned early to treat every spill as a team exercise—solo fixes lead to mistakes, so bring a second pair of eyes.
Training and Communication Make the Difference
No safety procedure matters if teams skip the training. Refresher courses save lives because people forget—even the most experienced chemist. Posting emergency numbers on every bench and holding regular walk-throughs of procedures keeps details fresh. I’ve watched newer techs ask for help when in doubt, which makes me trust our system more than any binder full of safety protocols.
The Small Things Save Lives
Sometimes people joke about “paranoid” lab behavior, but in the case of tert-Butyl peroxymaleate, caution becomes community care. Every label, glove, and shield isn’t about bureaucracy—it’s about the stories told by those who didn’t take these steps. Don’t rush. Don’t work alone. And never treat peroxide safety as just another box to check.
Digging Into the Chemical Formula
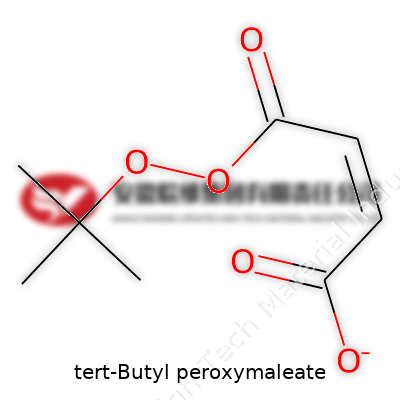
Understanding chemical structures means more than just memorizing letters and numbers. For tert-Butyl peroxymaleate, the formula is C8H12O6. This compound blends the features of a tert-butyl peroxide group with a maleic acid core. Structurally, it connects tert-butyl peroxide (a well-known radical initiator) directly to the maleate portion, which gives it unique reactivity.
Why Chemical Structure Matters in Everyday Life
When I first studied organic chemistry, complex names like tert-Butyl peroxymaleate intimidated me. On closer look, these compounds turn up everywhere—in plastics, paints, and even medical materials. Peroxides like this one play a major role as initiators for polymerization. Basically, they help build up long plastic chains from small molecules, all by creating reactive radicals when exposed to heat or light.
Over the years, I’ve seen chemical suppliers and manufacturers lean toward specific peroxides because they offer tailored reactivity and predictable performance. tert-Butyl peroxymaleate splits cleanly at a moderate temperature, so it frees up radicals without too much fuss. This makes it valuable for systems needing reliable and consistent initiation, such as in food packaging materials or medical devices that can’t risk contamination or residue.
The Real-World Impact: Safety and the Environment
Safety comes up every time peroxides are mentioned, and for good reason. Organic peroxides can break down and release oxygen, turning a stable warehouse into a potential accident scene if mishandled. This compound is no exception. Handling it with respect—proper temperature control, away from metals, and using explosion-proof equipment—matters just as much as getting its formula right.
Workers on the ground know that many peroxides are incompatible with common materials found in factories. Accidental mixing can spark fires or explosions. These risks pushed many companies to train staff regularly and update safety sheets, saving lives and avoiding massive property damage.
What Needs to Change for a Safer Future?
One big issue I’ve noticed is that knowledge does not always translate from laboratory books to industrial settings. Chemists understand the decomposition paths and hazards, but warehouse operators or factory technicians sometimes only remember the broad strokes. That’s a gap worth closing. More hands-on training, refresher sessions, and real-life drills—those efforts make the biggest difference in safety records.
Environmental concerns also drive research into alternatives. The breakdown products of peroxides sometimes linger in soil or water, raising regulatory and ecological red flags. Switches to less persistent, more biodegradable options help, but such changes do not happen overnight. Open communication with environmental regulators and communities around chemical plants helps rebuild trust, especially in regions where chemical use grew faster than oversight.
Staying Up to Date with Science
As more industries demand sustainable chemistry, formulas like C8H12O6 invite new research. Chemists in academia and industry constantly review reaction mechanisms, decomposition rates, and ways to tune reactivity or minimize hazards. Events like international accidents serve as harsh reminders that complacency proves costly. Continuing education, deeper safety culture, and a willingness to innovate offer the best path forward for everyone working with these chemicals.
Understanding the Risks
tert-Butyl peroxymaleate isn’t something you leave on a lab bench and forget. Over the years, I’ve seen more than one rushed chemist find out the hard way how unforgiving these organic peroxides can be. Working around unstable chemicals reminds me why safe storage isn’t optional. This compound decomposes under heat, friction, or if it comes in contact with contaminants. Breakdowns like these can cause fires or explosions. Chemicals react with people, not just with other reagents, so policies around storage always deserve attention.
Temperature and Location Concerns
A cool, dry place, away from direct sunlight and heat sources, keeps tert-Butyl peroxymaleate stable. Room temperature may feel fine for sugars or salt, but not for this material. Most manufacturers recommend temperatures below 30°C, some err lower. Warm storerooms or improvised closets near radiators risk trouble. I always aim for dedicated chemical storage units, often lined with trays to catch possible leaks. Avoiding wooden shelving makes sense, since wood can fuel a fire. Industry guides suggest stand-alone storage, never mixed with flammables, reducing the risk of chain reactions during an incident.
Separation From Incompatibles
I remember a case where a technician stored an oxidizer too close to metal powders. That mistake led to a scary cleanup. tert-Butyl peroxymaleate belongs far from reducing agents, metal salts, or acids. Contact with any of these kicks off hazardous reactions. Segregating peroxides from common solvents, paper supplies, and glass cleaners isn’t just paperwork, it prevents disasters. When chemicals remain in original containers with clear labels, mistakes drop sharply.
Choosing The Right Containers
Only containers built for peroxides get my vote. Manufacturers supply tert-Butyl peroxymaleate in tight-sealed, vented, and UV-resistant bottles. I’ve seen accidents where chemicals moved into unlabeled jars. That never ends well. If someone needs to transfer material, printed hazard symbols and date markings halt confusion. Older peroxides can form crystals at the lid; never force these open. Trained staff with the right tools and safety gear can handle these, and regular audits keep everyone sharp.
Fire Protection and Ventilation
Automated sprinklers and fire-rated cabinets, along with smoke detectors, turn worst-case events into stories rather than front-page news. Dedicated ventilation draws away fumes, cutting exposure risk. Eye washes and emergency showers installed nearby show respect for anyone working in these rooms. Training makes a real difference—everyone from lab techs to janitors benefits from clear instructions and drills.
Legal Rules and Documentation
Regulators set limits for storing organic peroxides. Local and national codes sometimes set strict weight or volume caps for facilities and transportation. Every time I see a chemical spreadsheet, I check that logbooks earmark expiration dates and batch numbers. Hazardous waste procedures play a role: nothing beats good paperwork alongside reasonable quantities on-site. If storage outlasts the shelf life or there’s ever doubt about stability, disposal by professional teams is best.
Raising Standards, Saving Lives
Complacency has no place near tert-Butyl peroxymaleate. Risk grows with time, weather, and sloppy habits. Routine checks, clear labels, and respect for incompatibles keep everyone safer. When facilities take care in these ways, incidents stay rare. In my experience, reacting calmly and methodically beats frantically googling safety rules in a crisis. Proper handling and storage don’t just tick a box—they protect health, property, and reputations.
Understanding Stability: Why It Matters
Chemicals live in a world that rarely forgives mistakes. In a laboratory, a warehouse, or on the factory floor, the stakes ride on the actual properties of each compound, not just numbers in a brochure. Tert-butyl peroxymaleate fits that bill. It’s a workhorse in polymerization, where even a tiny shift in reactivity or purity changes the product down the line. Relying on fresh, stable materials pays off. If a shelf life gets ignored, technicians risk fire, inconsistent batches, or failed quality inspections. For me, the lesson has always been simple: treat chemicals with as much respect as possible, because the molecule won’t forgive a sloppy calendar.
Shelf Life Facts and Industry Practice
Tert-butyl peroxymaleate doesn’t break from the pack of other organic peroxides in how it ages. Its shelf life usually falls between six months and a year if managed with strict adherence to safety and storage rules. Decomposition speeds up with warmth, sunlight, or exposure to moisture. Keep it in a cool, dry spot and the clock ticks slower. The companies I’ve worked with always demanded temperature logs for every fridge or storage area housing peroxides. Any blip above recommended temperatures led straight to a risk analysis. Some storage managers install redundant alarms against heat spikes for just this reason.
Regulators like OSHA and ECHA don’t just set rules out of the blue. These guidelines build from fire safety history and years of chemical testing. tert-Butyl peroxymaleate, with its oxygen-rich structure, demands respect. Old stock can destabilize, meaning surprise events from pressure build-up or, far worse, an explosion. Most suppliers print expiration dates based on valid stability tests, which mirror actual conditions where customers use the stuff. I’ve seen audits go south when expired lots sneak past oversight. Following shelf life guidelines doesn’t just keep the workplace safe—it prevents expensive and public mistakes.
Risk Management and Quality Control
Ignoring shelf life short-changes everyone. If you use decomposed tert-butyl peroxymaleate, a polymer process may drift off-target, wasting days of effort. After seeing batches thrown out, with teams picking through every possible cause, it’s hard to overstate the importance of basic chemical discipline. Good operations keep digital logs of every shipment. Periodic re-testing on stored lots, as a backup, has saved my teams from headaches when supply chains stall.
Storage matters as much as paperwork. Some try to extend shelf life with refrigeration, but condensation invites its own hazards. Aerosolized peroxide, caused by temperature swings or cracked containers, can spread danger beyond the chemical store. Handling rules—grounding, proper venting, and not stacking containers—save lives. I once watched a near miss as a leaking drum, too old and sitting near a heater, released vapor that could have gone badly in a busy shop. Don’t cut corners.
Smart Solutions for Real-World Work
More labs and factories automate their inventory reminders so outdated peroxides never linger out of habit. Revising purchase orders more often shrinks the window for trouble. Relationships with suppliers come into play; a reliable partner shares up-to-date technical data, and honest shelf life statements, and works with you on returns for outdated material. I always advise staying skeptical of discount bulk purchases if storage isn’t tight and turnover is slow. No savings cover a chemical incident or ruined production run.
Respecting the shelf life of tert-butyl peroxymaleate isn’t just good science—it’s the bedrock of safe, consistent production. The lessons come from the field, not from textbooks: use chemicals well within their use-by dates, store them right, and trust experience just as much as paperwork. Mistakes can be loud, expensive, and dangerous. Good habits make the difference.