tert-Butyl Peroxybenzoate: A Deep Dive into Its Story and Impact
Historical Development
The discovery of tert-butyl peroxybenzoate traces back to a time when organic chemists wanted new ways to spark reactions at lower temperatures and with greater control. In the 1950s, the search for safer, more stable peroxides put a spotlight on organic peroxy esters. Traditional initiators for polymerization—such as benzoyl peroxide—had their uses, but researchers recognized tert-butyl peroxybenzoate brought a useful mix of thermal stability and reactivity. Over the decades, large and small chemical companies tinkered with its synthesis, looking for higher yields and fewer side-products. The gradual shift from batch processes toward continuous production lines in the chemical industry improved quality and safety. Tert-butyl peroxybenzoate eventually found a home in a variety of applications, thanks in large part to decades of incremental advances in organic peroxide chemistry and a series of regulatory changes that favored less hazardous substances. These developments highlight the way necessity and technical know-how combine to shape the chemical landscape.
Product Overview
Tert-butyl peroxybenzoate, often shortened to TBPB or traded under names like Trigonox C, presents as a colorless or slightly yellowish liquid with a distinctive, pungent odor. This chemical functions mainly as a radical initiator. Manufacturers in the plastics and resins industry depend on it to kickstart and control polymerization. In my own experience with industrial materials, the sheer reliability of TBPB stands out. The product’s balance between reactivity and storage safety reduces headaches over waste and spoilage. Its compatibility with several monomers—including styrene and acrylics—places it on the go-to shelf for commercial users. Even small research labs use it for making specialty polymers and elastomers, though large reactors use far more to create tons of industry-standard plastics.
Physical & Chemical Properties
Chemists look at TBPB and see a substance weighing in at 206.26 g/mol, with the formula C11H14O3. The liquid’s boiling point sits around 150-152°C under reduced pressure. It separates out from water and prefers the company of organic solvents like acetone and toluene. At room temperature, TBPB keeps its cool, but once heated above 85°C, things get moving fast. The oxygen-oxygen single bond in the perester group holds enough stored energy that, under the right circumstances, it breaks apart, launching reactive radicals that push reactions forward. Handling it without proper controls can lead to unpleasant surprises: its autoignition temperature sits at about 260°C, and it will decompose violently if left in hot spots. I’ve seen shops where a fridge dedicated to peroxides, including TBPB, sits padlocked for good reason. Its density hovers near 1.06 g/cm³, and it carries a vapor pressure of 1 mmHg at roughly 20°C—enough to give off vapors that need respect in confined spaces.
Technical Specifications & Labeling
A typical drum or container of tert-butyl peroxybenzoate comes labeled with the UN number 3109, flagging it as an organic peroxide type E, liquid. Labels display hazard pictograms for explosive and flammable substances, plus strict handling and storage instructions. Global Harmonized System (GHS) regulations require manufacturers to attach detailed safety data sheets, complete with recommended PPE, emergency procedures, and disposal strategies. Most technical grade TBPB offers purity above 98%, with tight control on acidity, water, and stabilizer content. Certificate of analysis reports outline the peroxide value—measuring oxidizing strength in grams per 100 grams of sample—which ensures the end-user can trust the lot’s performance. Each drum usually includes manufacture date, batch number, recommended storage temperature (often 2-8°C), and shelf life, usually capped at 12 months for best results. Accurate and thorough information upfront supports quality assurance from the lab bench to the production plant.
Preparation Method
Producing tert-butyl peroxybenzoate usually starts with the reaction of benzoyl chloride with tert-butyl hydroperoxide in the presence of a base. Some manufacturers use sodium carbonate or pyridine to soak up the hydrogen chloride released during the reaction, steering it away from corroding equipment or contaminating the product. Process engineers must control temperature and reactant ratios carefully, as both overheat and excess reactants can lead to runaway reactions or poor product yield. After the chemical dance finishes, the mixture goes through separation, washing, and vacuum distillation steps to remove impurities and stabilize the final product. Drawing on past experience, I recall how even the smallest traces of metallic residues or acid can spark decomposition, so production lines often involve stainless steel reactors lined with inert coatings. Waste byproducts demand careful neutralization and disposal, not just for environmental safety but to stop accidental fires or explosions. Scaling up this synthesis for commercial output challenges operators to balance yield, quality, and safety in every shift.
Chemical Reactions & Modifications
Tert-butyl peroxybenzoate sits in the toolbox of free-radical chemistry. Its core strength lies in homolytic cleavage under gentle heat, usually between 80°C and 125°C, which produces tert-butoxy and benzoyloxy radicals. Both play starring roles in kicking off polymerization or cross-linking of unsaturated polyesters and vinyl monomers. The precise initiation temperature can be tuned by mixing TBPB with other peroxides or using co-initiators, giving chemists greater control over reaction rates and final material properties. Reaction byproducts, mainly tert-butanol and benzoic acid, can often be removed or utilized, making processes more sustainable. Modifying TBPB itself takes skill; substitution or structural tweaking can yield peresters with shifted reactivity or improved safety profiles. In some custom applications, researchers add stabilizers or phlegmatizers to smooth out the energy release, especially in large-scale batches where heat transfer can build up fast. Each tweak in process or formulation brings subtle shifts in downstream behavior, reminding chemists that a successful result depends on mastering the details.
Synonyms & Product Names
Tert-butyl peroxybenzoate appears in catalogs and technical papers under a stack of names: TBPB, Trigonox C, Luperox P, Perkadox OB, and benzoyl peroxide butyl tert-ester. Some labels might call it tert-butyl perbenzoate or list international codes like EC No. 201-279-3 and CAS No. 614-45-9. The language can get tangled, so industry insiders rely on chemical structure and purity specs to avoid confusion. Each synonym echoes a supplier’s branding or historical naming convention, but the core molecule and function remain the same. From experience, cross-checking registry numbers and supplier data goes a long way to cut through the clutter, especially for procurement and regulatory compliance teams managing global inventories.
Safety & Operational Standards
Handling TBPB isn’t for the inexperienced. Organic peroxides call for routine, careful protocols, and TBPB’s inherited hazards—flammability and explosive decomposition—raise the stakes. Safety standards, like those from OSHA and REACH, specify full-face shields, chemical-resistant gloves, lab coats, and explosion-proof hoods. Special care goes into segregating TBPB from acids, other peroxides, and strong reducing agents. In production settings, temperature controls with redundant fail-safes ward off accidental heating, and continuous monitoring catches vapor leaks before they get out of hand. Fire suppression systems must be ready and compatible; water can spread a peroxide fire, so dry powder or CO2 systems see regular use in these areas. Storage lockers labeled with prominent signage typically sit apart from populated workspaces, caged and ventilated, cooled to stay below the critical self-accelerating decomposition temperature. Long-term workers know the importance of regular safety training and emergency drills, which can spell the difference between a close call and a disaster.
Application Area
TBPB acts as the backbone for many manufacturing processes. The plastics industry depends on its ability to trigger polymerization for resins, especially acrylics, polystyrene, and unsaturated polyester materials found in fiberglass, boat hulls, and automotive parts. Even adhesives and coatings benefit from TBPB’s steady, predictable radical output, driving performance that meets rising consumer and regulatory expectations. In the rubber sector, cross-linking steps depend on TBPB to give tires and seals their mechanical strength. Some pharmaceuticals and specialty fine chemicals capitalize on TBPB’s selectivity in specific oxidation reactions. Small R&D labs, along with global players, all find a role for TBPB, whether developing impact-resistant plastics or tuning the weatherability of paints. From the rolling lines of an automotive plant to benchtop experiments testing new composites, TBPB keeps work moving forward, underscoring the link between specialty chemicals and everyday products.
Research & Development
Chemists and engineers in research push TBPB’s boundaries every year. Some of the most promising work seeks to combine TBPB with renewable feedstocks to make safer, greener plastics. Others investigate how trace additives can suppress unwanted side reactions or enhance shelf-life. Computational chemists use simulation tools to predict how changes in TBPB’s structure might affect decomposition rates and product profiles. Analytical labs invest in advanced monitoring—infrared and NMR spectrometers, gas chromatographs—to watch TBPB behavior during real-world reactions, seeking better process control and troubleshooting hidden inefficiencies. Collaborative projects mix the talents of industrial veterans and academic innovators, creating a pipeline for new uses or improved safety approaches. From a personal perspective, the spirit of shared problem-solving drives much of the progress: researchers talk across company lines at conferences, trade technical notes, and train the next generation of scientists who will inherit both TBPB’s benefits and its risks.
Toxicity Research
Tert-butyl peroxybenzoate’s health impacts demand close attention. Acute exposure, often through inhalation or prolonged skin contact, leads to irritation and sensitization. Animal studies reveal that high doses, often above those encountered in standard workplace settings, can overwhelm the liver and kidneys. Chronic exposure remains less well characterized, but experiences in chemical plants led to tight exposure limits and detailed handling guidelines. TBPB breaks down into tert-butanol and benzoic acid, both better researched for environmental and health effects; neither is free from concern, especially in aquatic systems. European and American regulators periodically review toxicity data, pushing for lower allowable limits as new findings emerge. I’ve encountered strict routine health monitoring programs for anyone working with TBPB, including regular blood tests and air monitoring in production rooms. These safeguards reflect a clear message: progress in performance must not come at the expense of worker health or environmental safety.
Future Prospects
Looking to the future, TBPB’s story isn’t over. Research into safer and less toxic radical sources could shift its dominance, but for now, the demand for precision and reliability in polymerization supports its place in the market. New regulations and green chemistry trends push producers to cut residual byproducts, improve shelf-life, and boost recycling of both product and packaging. Advances in process automation and real-time safety monitoring offer ways to mitigate the ever-present risks of peroxide use, opening opportunities for smaller producers and less centralized facilities. Companies that blend deep technical understanding with a commitment to human and environmental safety will shape TBPB’s legacy. Every tweak in manufacturing or application rests on decades of trial, error, and shared expertise, showing how something as precise as a perester molecule continues to evolve alongside the industries that depend on it.
A Closer Look at What It Actually Does
tert-Butyl peroxybenzoate, known in labs and factories as TBPB, has long played a unique part in chemical manufacturing. It acts as an initiator, which means companies rely on it to kickstart key reactions—especially those needed to make plastics and resins. Without these initiators, some of the strongest materials in our daily lives—think of outdoor furniture, medical devices, or even the insulation in your house—wouldn’t exist in their current form.
In my experience working alongside polymer engineers, TBPB showed up as the backbone of productivity in resin fabrication rooms. Factories love TBPB for making unsaturated polyester resins. You usually find these resins where a mix of strength, flexibility, and weather-resistance matters. For example, boat hulls, car panels, bathtubs, and surfboards all benefit from the properties this compound helps create.
Why Industry Depends on TBPB
The practical reason TBPB earns so much use comes down to its effectiveness at starting chain reactions at manageable temperatures. Some initiators demand high heat, which can raise costs or create safety hazards. TBPB lets technicians start curing resins at much lower temperatures, giving more control over the process and keeping energy costs in check.
Workers I’ve spoken with feel safer handling TBPB compared to some older peroxide initiators, which were far more sensitive to heat or shock. Most manufacturers now design their processes around safety data and regulations—the drive for safer and more predictable workflows gives TBPB another edge.
Environmental and Health Questions
There’s a conversation taking place about any industrial substance, and TBPB is no exception. Fact sheets point out that peroxides can present hazards if mishandled—skin contact, inhalation, or improper mixing can all pose real risks. Environmental researchers worry about what happens when TBPB production and use release residues or byproducts into waterways or the soil.
The good news rests on transparency and strict safety practices. Chemical plants invest heavily in training, protective clothing, and containment systems. Some countries ramp up audits and require detailed documentation on how chemicals like TBPB get used, tracked, and disposed of. In most modern workplaces, regulations align with best practices backed by science, not just habit.
Room for Progress and Sustainable Approaches
There’s always pressure on chemical producers to find cleaner alternatives or design products that break down more safely at the end of their lifecycle. TBPB’s manufacturers fund research looking into greener peroxides that break down more easily in the environment or involve less energy in their production. Some research teams have started exploring catalysts that can replace peroxides or at least reduce the quantities needed for each batch of resin or plastic.
Anyone invested in the future of manufacturing needs to keep pace with eco-friendly innovation. The big companies set aside funds to upgrade equipment that captures vapors and reduces waste. Workers, too, play a part—they voice safety concerns and keep management honest about best practice. With industry routines changing year by year, there’s room to keep chemicals like TBPB useful and safer for everyone involved.
Understanding the Risks
tert-Butyl peroxybenzoate often shows up in labs that work with polymerization or specialized organic syntheses. The trouble starts with its structure—the stuff’s an organic peroxide, so it can decompose, release heat, and kick off some wild reactions once pushed past certain limits. People sometimes underestimate how easily these kinds of materials can go from useful to dangerous. I’ve learned to take organic peroxides seriously after hearing about an accident where a bottle ruptured in a lab fridge because someone ignored storage rules. Nobody wants burning peroxide on their hands or an explosion close by.
Personal Protection Always Comes First
I always grab a long-sleeve lab coat, solid nitrile gloves, and goggles before lifting the lid on any container with tert-Butyl peroxybenzoate. Regular glasses make a poor substitute—splashes of this chemical won’t care that your eyewear looks professional. For larger quantities, I rely on a face shield, because splashes do happen. Skin contact shouldn’t get brushed off. If even a drop lands on you, fast rinsing beats bravado every time. The same goes for breathing: a fume hood draws away vapors that can sting your lungs or eyes, so don’t settle for a bench-top corner if you can help it.
Smart Storage Saves Lives
tert-Butyl peroxybenzoate prefers a cool, dry place—usually a chemical refrigerator dedicated to organic peroxides. Storing it with other flammable materials or acids can turn a small mishap into a disaster. I label every bottle with bold signage and always check the date, since peroxides get less stable over time. Locking storage helps, too—one stray bottle shouldn’t end up in someone else’s experiment by mistake. Never transfer the chemical into random containers; always use original bottles or ones approved specifically for peroxides. That little detail helped a colleague avoid a near miss when an intern almost grabbed the wrong solvent for cleaning.
Preventing Spills and Fires
Peroxides like this react badly with heat, sunlight, friction, and sparks. I cut out all unnecessary ignition sources and use tools that don’t shed static. My workspace stays uncluttered—no papers, oily rags, or other combustibles near the action. When weighing or transferring, I use slow, careful motions. If a spill occurs, I use absorbent pads and cleaning solutions rated for peroxides. Regular paper towels or mops don’t cut it—they can cause a reaction. Keeping a suitable fire extinguisher nearby—foam or dry powder—keeps panic down if things go sideways.
Disposal: No Corners Cut
Disposing of tert-Butyl peroxybenzoate means following solid local regulations. I never dump leftovers down the drain, since water and sewage systems can’t handle this type of compound. Waste must go in a designated container with a tight-fitting lid, clearly labeled for hazardous pickup. One mistake I’ve seen: storing waste peroxide with other organic waste by mistake. A nasty decomposition caught just in time. Training others to spot differences in labeling and container types helps prevent these mix-ups.
Learning and Training
Written procedures make a huge difference. Clear posters, regular reminders, and real-world examples stick better than lectures. If you’ve never worked with peroxides, team up with someone who knows their tricks. Reporting and learning from near misses gets everyone home safe, and no step is too basic when safety is on the line. It’s not about being afraid of chemicals; it’s about taking their power seriously.
The Real Risks of Mishandling This Chemical
Working in chemical manufacturing has taught me how one simple lapse in storage can mean chaos. tert-Butyl peroxybenzoate sits among those chemicals that seem harmless until they aren’t. Every time I see a dented drum or faded label, I remember the stories—small leaks that ended with frantic evacuations and blown insurance claims. This isn't just a fancy name on a shipment manifest. It presents real danger if forgotten in a warm corner or cracked container.
Oxidizing Power Demands Respect
This chemical acts as a strong organic peroxide, ready to decompose and release gases—sometimes with a lot of heat. Once temperatures start creeping much over 25°C (77°F), decomposition picks up. Excess heat transforms stable product into a runaway reaction, which can mean fire or even explosion in closed spaces. For folks who haven’t worked with peroxides before, here’s the most important thing: they work as catalysts but also have a mind of their own when ignored.
Storage Details That Actually Matter
The safest spot keeps tert-Butyl peroxybenzoate cool, well away from sunlight and sources of ignition. I always look for dedicated peroxide storage rooms, set apart from main traffic and other chemical storage. A fridge or cold room set around 2-8°C (36-46°F) discourages any urge the chemical might have to break down early. This is not the place for a makeshift shed or a corner in the maintenance closet. Nothing beats a storage zone with proper ventilation—enclosed spaces will trap any vapors, raising risk as soon as something leaks.
Labeling earns respect here too. Without a clear hazard symbol—maybe a freshly printed “Oxidizer” and “Organic Peroxide Type C”—too many people might treat this like just another bottle of solvent. I’ve had coworkers thank me after we caught a shipment without the right symbols, before something went wrong.
Containers and Compatibility
Keeping tert-Butyl peroxybenzoate safely sealed means high-quality containers, often polyethylene with screw lids—no glass with stress cracks, no rusty metal, no jerry cans used for anything else. Corrosive breakdown eats into containers over time, especially if stored too long without inspections. I schedule checks every two weeks. Every shift supervisor can set up reminder routines. Don’t let containers sit past their expiration dates, and resist the urge to transfer into smaller bottles for “convenience.” Spills tend to happen in moments like that.
Segregation: Preventing Accidental Mixtures
Chemical neighbors make a difference. tert-Butyl peroxybenzoate doesn’t play well near acids, bases, reducing agents, or regular combustibles like paper and rags. Close calls happen during big clean-ups, when people toss unrelated waste into peroxides’ designated bins. Training staff to understand why separation matters—backed up by stories, not just rules—cuts down on these risks.
Emergency Readiness Isn’t Optional
Treat safety showers, eyewash stations, and spill kits as part of the storage package. I found that safety drills catch more attention when you describe real mistakes and show how a spill kit cuts panic in half. Emergency procedures, practiced twice a year, burn into memory. Updated Material Safety Data Sheets stay taped to the inside door—no internet search needed if someone’s eyes burn or a cloud rises out of a drum.
Building a Strong Safety Culture
Once, complacency felt tempting—a well-run shop, zero past incidents, good reviews from inspectors. Then an unexpected power outage left cold storage rooms warming up. The backup alarms saved us, but nobody in that warehouse forgot the close call. tert-Butyl peroxybenzoate rewards those who never take shortcuts. Safety grows out of habits, clear communication, and a willingness to ask the next person, “Does this look right to you?” Experience makes the rules, but people keep everyone safe.
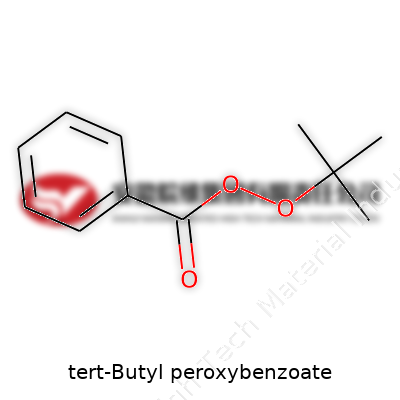
Breaking Down the Molecule
Picture a puzzle—every atom fits into a specific spot for the whole thing to work. That’s what goes on inside tert-Butyl peroxybenzoate. This compound, with the formula C11H14O3, plays a big role as a radical initiator in the plastics and polymer world. Its structure brings together a benzoate group and a tert-butyl group, linked through a peroxide bond—a weak link that makes this compound so reactive and useful.
Starting with the backbone: the benzene ring, six carbons locked in a hexagon, brings aromatic stability. Connected to this ring is a carbonyl group (C=O), which forms the benzoate part. The oxygen next to it forms a bridge, linking to the tert-butyl group. This is where things get interesting—the central O–O bond, known as a peroxide linkage, makes the chemical sensitive to heat, light, and shock. The tert-butyl side, shaped almost like a three-legged stool with three methyl arms branching from a central carbon, is bulky and highly branched. Energy arrives, that peroxide bond splits, and free radicals burst onto the scene.
Why the Structure Matters in Real Life
Working in the lab, I’ve seen the importance of molecule shape firsthand. You can’t swap out the tert-butyl group for just anything—the size helps control when and how fast the bond breaks. Industries rely on this trait to kick off polymerization at the right temperature. If you use the wrong compound, you end up with polymers that don’t act the way you want, causing wasted time and money. Facts on the ground matter, and chemistry offers no shortcuts.
Safety can’t be skipped here. Those peroxide bonds don’t just break on command—exposure to heat or impact can turn a stable container into a dangerous situation real quick. You need storage away from sunlight and heat, with every lab tech drilled on handling protocols. According to OSHA and other safety agencies, improper management of peroxy compounds has led to explosions and major incidents. I remember one story from grad school: a poorly secured bottle of a similar compound led to an evacuation and days of decontamination.
Moving Toward Solutions
Manufacturers look for ways to ship and store these compounds more safely, like using stabilizers that slow decomposition under regular conditions. Chemists try tweaking the tert-butyl group or the benzoate part to make peroxides that still initiate reactions but with a lower risk profile. Consistent oversight and practical training make a difference—nobody should treat a peroxide compound like just any other bottle on the shelf.
Many companies now use smaller quantities or set up real-time monitoring in production lines. Sensors pick up temperature spikes right away, letting workers know if a batch is running hot. Sharing information across the industry makes a difference; mistakes don’t need to repeat just because a new person walks through the door. Regulators publish detailed guidelines for transport and labeling, so even a new tech can recognize danger just by glancing at a container.
Today, everyone from students to factory owners needs a firm handle on what makes tert-Butyl peroxybenzoate tick. A clear grasp of structure and reactivity can cut down on accidents, boost efficiency in production, and open doors to better, safer materials in the future. The devil’s in the details—and chemicals like this one prove it every day.
A Closer Look at Chemical Longevity
tert-Butyl peroxybenzoate helps push countless industrial processes across various sectors, from polymer production to pharmaceuticals. Digging into its shelf life isn’t a lab-only concern — it speaks to safety, efficiency, and even bottom lines.
Status of Shelf Life: Facts and Realities
Manufacturers state that tert-butyl peroxybenzoate typically holds up for around twelve months under proper storage. That figure comes with a simple premise: keep the substance cool, dry, away from direct sunlight and heat sources. Temperatures above 30°C can cut that shelf life in half. I’ve seen companies place their stocks in temperature-monitored storage and keep detailed logs, just to stay on top of this.
Let the heat creep in, and things get risky. This compound decomposes, builds up pressure, and at the worst, may turn hazardous. The United Nations ranks it as a “Class 5.2 organic peroxide,” so no one in their right mind shrugs off storage rules. Decomposition not only means dangerous byproducts but also hits the reliability of any process using degraded material.
Why Shelf Life Shortens
Peroxides break down over time, especially if handled loosely. Any exposure to moisture or oxygen — even from a briefly opened cap — takes a bite off that twelve-month standard. Humidity affects the rate of breakdown. I’ve known quality control engineers who run spot-tests every few months, just to avoid placing old or suspect batches into production. One poor batch with lost reactivity can spark a costly recall or bottleneck.
Labeling often gets overlooked, yet I’ve watched mistakes happen at plants where staff missed the expiry date or batch number. Outdated stock winds up in the mix, and all sorts of regulatory and quality issues kick in. Responsible teams audit their stockrooms and keep clear labeling front and center to keep those headaches at bay.
Safety, Waste, and Accountability
The shelf life matters beyond just quality. Authorities like OSHA and the EPA lay down strict rules about handling and disposal. Outdated tert-butyl peroxybenzoate becomes hazardous waste. Disposing of it properly means documentation, transportation by certified handlers, and steep fees — something plant managers try to avoid by using up valid stock before it hits waste status. In practice, this encourages smaller, more frequent shipments or “just-in-time” supply strategies.
The cost of waste can pile up, especially for users with slow turnover. Smaller operations sometimes share inventory or coordinate bigger bulk orders to minimize product idle time. I’ve worked with purchasing agents who rely on tight relationships with chemical suppliers to keep stock moving at just the right pace.
Staying Ahead: Proven Approaches
A smart storage plan forms the backbone of safe and cost-effective tert-butyl peroxybenzoate management. Regular audits, staff training, and strict adherence to manufacturer specifications make the difference. Some facilities introduce automated inventory systems with alerts tied to expiry dates, cutting out human error and reducing risk. Audits help, but nothing beats a well-trained team who understands what’s at stake. Safe, responsible use starts at the warehouse door, and attention to shelf life turns a potential hazard into an everyday workhorse.