tert-Butyl Peroxyacetate: Far from Obscure Chemical, Central to Modern Industry
Historical Perspective: A Glimpse into Development
The move toward safer and more predictable organic peroxides traces a clear path back to the middle of the twentieth century. Researchers started working with simple peroxides, searching for substances with high reactivity, lower volatility, and manageable hazards. tert-Butyl peroxyacetate came to life as breakthroughs in organic synthesis and polymer technology pushed industries to demand new tools. Scientists learned that dialkyl peroxides provided control and predictability, leading them to this unique compound. As polymer chemists tinkered with new methods, they realized that adding tert-butyl peroxyacetate brought both efficiency and reduced risk compared to earlier, more reactive peroxides. Fast-forward to today, and this compound doesn’t only hold historical importance—it keeps forming the backbone for polymer modifications and safety improvements in many manufacturing floors.
Product Overview: Role in Modern Processes
Picture a clear, oily liquid with a strong smell, packed into sturdy steel drums or HDPE containers. tert-Butyl peroxyacetate stands out not for its glamour, but for reliability. Industrial workers turn to it for polymerization of vinyl chloride and styrene, knowing it’ll deliver cleaner reactions and more predictable molecular weights. Chemists see it as a solid choice—not as showy as some specialty catalysts, but one that keeps accidents at bay and products consistent. As companies refine their processes for automotive parts, coatings, or latex production, this peroxide shows up in production lines where mistakes mean wasted time, stricter regulations, or worse, a hazardous event.
Physical & Chemical Properties: What Sets It Apart
tert-Butyl peroxyacetate has a molecular structure that looks deceptively simple, but its performance reveals depth. It holds a moderate boiling point, usually between 90 and 130°C, which keeps it workable in common equipment designed for organic peroxides. A flash point close to room temperature means you must pay attention to storage and handling—even short lapses in safety can result in dangerous vapor releases. Its solubility in many organic solvents allows easy mixing into commercial resins. There’s a solid reason manufacturers keep data sheets in every plant—it decomposes steadily at elevated temperatures, producing radicals that drive chemical transformations. Again and again, analysts revisit its chemical structure, seeing the tert-butyl group and the peroxyacetate moiety as a combination that resists unwanted side reactions while still providing the trigger for big polymer changes.
Technical Specifications & Labeling: Getting It Right
Anyone who’s ever unloaded a drum of tert-butyl peroxyacetate would recognize the bright warning labels and standardized hazard codes. Regulatory bodies require detailed information: UN numbers, hazard statements, pictograms for flammability and oxidizing potential, and rigorously checked concentration ranges—often 75% active ingredient in a phlegmatizer to “tame” its reactivity. Labeling isn’t a bureaucratic hoop; it saves lives. Not all chemical suppliers use the same stabilizers, so knowing the exact formulation helps emergency responders and plant operators. Fact sheets list precise melting and decomposition points. Larger users rely on certificates of analysis—not only for regulatory compliance, but because batch-to-batch consistency affects their end product properties, from vinyl windows to synthetic rubbers.
Preparation Method: No Shortcuts, Just Chemistry
Making tert-butyl peroxyacetate starts with careful reactions between tert-butyl hydroperoxide and acetic anhydride or acetyl chloride. Anyone who’s watched these reactions in a lab sees the exothermic response—temperature control becomes non-negotiable. Without that, the process becomes a runaway risk. Reactor design uses glass, stainless steel, or lined vessels to resist corrosion and keep leaks in check. Skilled technicians cool the mixture as the acetylating agent reacts with the hydroperoxide, separating phases to avoid any build-up of peroxides. Purification usually involves distillation under reduced pressure, followed by careful stabilization with small amounts of phlegmatizers, so the final product ships safely without degradation during transit.
Chemical Reactions & Modifications: Powerhouse of Radical Chemistry
tert-Butyl peroxyacetate owes much of its value to its stubborn tendency to break down in a controlled way at industrial polymerization temperatures, pumping out free radicals. Polymer engineers use these radicals to start chain reactions, transforming small monomers like vinyl chloride into durable plastics. In the lab, researchers have explored tailoring its decomposition temperature with additives, opening up new avenues to control polymer characteristics—flexibility, tensile strength, and even color fastness. The molecule’s stability compared to diacetyl peroxide, for example, lets users push for higher conversions in reactive resins or crosslinking of thermosets. Attempts to modify its backbone haven’t led to huge commercial success, but every year, a few research groups try blending in stabilizers to further improve its safety profile.
Synonyms & Product Names: Know Them or Risk a Costly Mistake
Listing off the full range of chemical synonyms can seem tedious, but it’s essential. Older safety sheets, customs documentation, and global regulatory filings might call it Acetic acid, peroxy-, 1,1-dimethylethyl ester, or t-Butyl peroxyacetate. In trade, it sometimes shows up under product codes from Arkema, United Initiators, or small specialty suppliers. Purchasing departments check these synonyms to avoid mis-orders—mistaken identity risks improper handling, incorrect storage conditions, or flawed polymer batches. Brand names vary, but every batch traces back to the same functional group, those tell-tale peroxide bonds.
Safety & Operational Standards: A Lesson Written in Experience
Anyone who’s worked with tert-butyl peroxyacetate learns respect for its hazards. Industry’s move toward safety after high-profile peroxide accidents cemented new handling standards: small-temperature margin between safe storage and dangerous decomposition; the need for shatterproof eye protection, face shields, chemical-resistant gloves, and remote filling equipment; no smoking or open flames anywhere near storage lockers. Emergency teams drill for vapor release scenarios. Regulations require peroxide storage in separate buildings away from organic materials or ignition sources. Operators rely on well-maintained venting systems and real-time temperature probes, not out of paranoia, but because history punishes complacency. When someone ignores a “minor” leak, the costs multiply rapidly.
Application Area: Staple of Industrial Polymer Chemistry
Whenever someone mentions the polymerization of polyvinyl chloride, polystyrene, or polyacrylics, tert-butyl peroxyacetate usually sits on the material list. People in the automotive industry need its initiator power to create flexible plastics for dashboards and trim. Paint and coating manufacturers appreciate how it controls curing of emulsions, regulating film formation and tack. Water treatment companies and paper mills take advantage of its solid reactivity profile in specialty resin applications, often after looking for more reliable alternatives to azo compounds. Newer lithium-ion battery technologies even mention it in notes about separator membrane production. It might not make headlines, but every time a consumer bends a plastic part in their hands or lingers near a new-car smell, they’re touching the result of this unassuming chemical’s work.
Research & Development: Chasing Innovation and Responsibility
Active labs across Asia, Europe, and the Americas look for ways to push tert-butyl peroxyacetate’s utility beyond the boundaries set in the sixties and seventies. Research teams work on tailoring its decomposition rates, blending with co-initiators, and finding new phlegmatizers that cut irritation or environmental degradation. Analysts in environmental chemistry study its fate after use—targeting reduction of residuals in effluent streams and designing catalysts that work at lower doses, reducing the chemical’s load in ecosystems. At the same time, process safety researchers develop new packaging to further cut explosion risk, labelling systems that communicate globally regardless of language, and personal protective equipment that responds to worst-case peroxide splashes. Less is left to chance now; research funds chase improvements that clear regulatory hurdles and help downstream industries stay competitive.
Toxicity Research: The Limits of Safety
Toxicologists don’t mince words about organic peroxides. tert-Butyl peroxyacetate irritates skin, eyes, and mucous membranes—direct contact typically leads to immediate burning and long-term problems if not treated. Chronic exposure links up with organ toxicity, so monitoring of production environments stays tight. Studies in animal models show central nervous system impact at relatively high doses. Regulatory agencies set tough occupational exposure limits, so manufacturers install local exhaust ventilation, enforce rotation of exposed personnel, and mandate annual health screens. Accidental releases put not just workers but nearby neighborhoods at risk, making containment protocols critical. There’s a trend toward greener replacements, but no perfect solution yet—the cost-performance ratio keeps tert-butyl peroxyacetate in broad use, even as research pushes for safer analogs.
Future Prospects: Beyond Industrial Routine
Tert-butyl peroxyacetate doesn’t rest on tradition. Rising demand for recyclable plastics and tighter emission standards trigger a wave of innovation. Companies explore blends that decompose at lower temperatures, letting manufacturers cut down on energy costs and greenhouse gas emissions. Novel uses in specialty adhesives and controlled-release agents hint at future growth. As environmental pressure increases, chemists look for formulations that degrade into less persistent byproducts—sometimes by altering the peroxyacetate backbone, sometimes by combining it with new additives. For now, its reliability and risk-management features keep it on order sheets, but the next few decades may see greener versions—or entirely new compounds—stepping in as plant managers and regulators demand stricter life-cycle oversight. Experience shows that science keeps moving, always chasing safer, smarter solutions in the world of industrial chemistry.
What Role Does tert-Butyl Peroxyacetate Play?
tert-Butyl peroxyacetate, often called TBPA, starts showing up in conversations about chemistry and manufacturing where things get complex and the stakes run high. This chemical doesn’t usually make headline news, but the story behind its uses says a lot about how modern materials get made. TBPA works as an initiator in polymerization, which is just a fancy way of saying it helps kick off the reactions that create plastics and resins. When I first started poking around the manufacturing floor years ago, I learned pretty fast that without the right starter, certain reactions either stall out or never launch at all. TBPA gives chemists a tool that’s both reliable and energetic, and manufacturers rely on it to meet tough production schedules and performance specs.
Why TBPA Earns Trust in Industry
This compound comes with a reputation for stability under transport and storage, which means fewer headaches for folks in supply chain and production. Its real edge, though, appears in its decomposition temperature, which lands squarely in a range favored for producing specialty polymers. If you’ve ever noticed how some plastics resist high heat or show off a glossy finish, there’s a good chance chemicals like TBPA played a part in setting those characteristics. In labs where every variable counts, engineers trust TBPA to start reactions exactly when needed, so batches remain consistent and expensive raw goods don't go to waste.
Looking at end uses, TBPA ends up influencing products from automotive coatings to adhesives. Car makers lean on durable polymers that come out of controlled reactions, and TBPA helps make those materials possible. In the electronics world, folks depend on polymers that can hold up against temperature swings and mechanical stress, and achieving that requires tight control over the polymer chains. TBPA doesn’t show up on the label, but the finished part relies on the clean chemistry behind it.
Safety, Sustainability, and Responsibility
From first exposure, I learned respect for chemicals like TBPA. Safety teams spend a lot of time managing material handling and training because TBPA, while handy, brings risks. It reacts strongly at the wrong temperature, so companies keep a close watch using sensors and clear protocols to cut down the chance of accidents. Over the years, facilities have adopted strict storage rules, fire suppression technology, and robust training. These standards come from hard lessons learned throughout the sector.
Sustainability teams now ask better questions about the legacy of these materials. Regulations such as REACH and frameworks in the US push manufacturers to think about lifecycle impacts. Waste management and emissions control deserve as much attention as reaction design. Some research efforts look to tweak peroxides for better biodegradability or lower volatility, hoping to ease the burden on downstream environments.
Where Do We Go From Here?
TBPA keeps industry moving, but the story wouldn’t stop with reliable polymerization. Innovation means keeping a careful balance between industrial productivity and community health. That balance takes clear safety standards, a willingness to invest in cleaner technologies, and honest review of every practice from procurement to disposal. Frontline workers, engineers, and managers—each group owns a piece of what happens in production halls every shift. The future for TBPA and chemicals like it depends on what those folks do next—how they adapt, what questions they’re willing to ask, and where they set the bar for performance and responsibility.
Respect the Risks that Come with this Chemical
tert-Butyl peroxyacetate, often found in industrial settings, can catch fire or explode without much warning. From personal lab experience, dealing with organic peroxides turns serious quickly. Ignoring a splash on your glove, leaving caps loose, or skipping a fume hood session could end in disaster. Factory workers and researchers have seen mistakes turn costly because somebody underestimated the risk or looked for shortcuts. This chemical won't forgive a casual mistake. It can give off dangerous fumes, and its vapors can travel to ignition sources even if you stay cautious with open flames. There’s no room for guessing when storing or transferring it—vigilance can make the difference between a safe day at the plant and a trip to the emergency room.
Proper Storage Keeps Everyone Safe
Storage for tert-Butyl peroxyacetate calls for attention to detail. Keep it in a cool, dry spot—away from heat, sunlight, or tightly-packed storerooms. Any careless stacking beside acids, paper, or cleaning chemicals creates more danger than most people realize. Explosions in plants sometimes trace back to overlooked details like incompatible storage or aging containers. Store it in original, approved packaging, away from direct sunlight or moisture that could start a runaway decomposition. Make sure security is tight; lock the area, train team members, and keep only the amount that’s needed for the job. Frequent walk-throughs with maintenance teams help catch leaks or swelling containers sooner rather than later. Records of shelf life help prevent old bottles from becoming ticking time bombs.
Preparation and PPE Save Lives
Personal protective equipment is more than just a checklist item—it’s a last line of defense. Lab coats rated for chemical resistance, safety goggles that seal, splash-proof gloves, and face shields all play roles. In the plant, people have tried to cut costs or move fast by reusing gloves, and sometimes, accidents make those shortcuts look foolish. Respiratory protection also deserves attention, not just for emergencies but for every transfer. Got ventilation? Make sure it’s running before opening the container. Engineers committed to real safety add automatic shutoff features and alarms in storage rooms—waiting until something goes wrong is too late.
Emergency Action Plans Make a Big Difference
Having a detailed emergency plan is not red tape—it’s what stops confusion from spreading during an accident. Employees should know who to call, where the eyewash and deluge showers are, and the evacuation routes. Real experience shows that drills, rather than handouts, stick in memory. Fire extinguishers rated for peroxides make sense near any storage site, and spill control materials should be close at hand—absorbent pads, inert clay, and sealed disposal drums. Relying on speed and instinct in a chemical emergency almost always goes poorly. Those who have faced leaks or small fires say keeping one’s head begins with good training and drills, not luck.
Prioritize Education and Reinforcement
Chemicals like tert-Butyl peroxyacetate demand ongoing education. Safety meetings feel repetitive until a near miss happens. Reading the SDS and pairing it with online courses—then testing for retention—shifts culture from compliance to real know-how. Scientists, engineers, and warehouse staff all benefit from training that digs into specific risks. In my years in chemical labs, cross-checking procedures and encouraging staff to speak up if something seems off have proved far more valuable than laminated policies. Supervisors who take time to explain “why” standards exist often see fewer accidents and better teamwork over time.
Everyday Chemistry at Work
tert-Butyl peroxyacetate looks like a handful of technical words. For me, growing up watching my dad work around engines and chemical drums in his workshop, that sort of name always brought a whiff of trouble and fascination. In the lab, it’s a regular on the shelf, not used as much as sodium chloride or acetic acid but still vital where it gets called for. Here’s the thing: every ingredient we use, from paint hardeners to advanced polymers, tells a story about risk, innovation, and a bit of long-haul thinking.
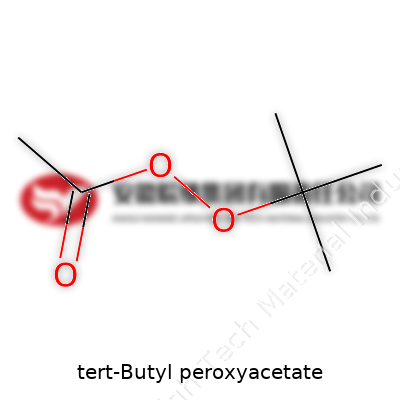
Getting to the Heart of the Formula
The chemical formula for tert-butyl peroxyacetate is C6H12O3. Those six carbons, twelve hydrogens, and three oxygens don’t look like much, but the real punch comes from the way they’re rigged together. You start with a tert-butyl group, a well-known carbon cluster—think of it as a crowded four-seater at a diner. Then you join it to a peroxyacetate group, which brings in the oxygen power. At its core, this molecule packs a peroxy bond (that’s an oxygen-oxygen single bond), and if you’ve ever mixed strong oxidizers, you know that’s a recipe for things happening fast—sometimes explosively so.
Why tert-Butyl Peroxyacetate Draws Attention
This chemical gets real use in polymer manufacturing. Factories use it to help make plastics through a process called polymerization. The peroxy bond in the molecule helps break apart big, sticky chains of molecules, so you don’t end up with brittle or useless material in everything from cell phone cases to medical devices. At the same time, anyone handling it learns quickly—this isn’t kitchen chemistry. The peroxy bond can break open at the wrong time and turn a quiet day into a mess. That’s why safety can’t be just a sign on the wall. Proper storage with temperature controls, real training, and regular drills are not just good ideas—they’re life-saving habits.
Safety in the Real World
Regulatory agencies like OSHA and the European Chemicals Agency track substances like this for a reason. Workers in the industry, and quite a few high school chemistry teachers, share stories—sometimes warnings—about chemical burns or accidental reactions. The risks are real, but they’re not a mystery. Good practices, solid labeling, and controlled environments keep the odds in favor of progress, not disaster. The rise of social media videos of experiments gone wrong underscores how easily a lack of focus or a skipped step can undo weeks or months of careful planning.
Smart Approaches for Tomorrow
Tert-butyl peroxyacetate’s value isn’t just as a catalyst. It prompts the bigger lesson: smart chemistry means respecting what molecules can do—and can undo—if you underestimate them. My own days in the lab taught me that shortcuts only look smart before something goes wrong. Clear protocols, ongoing safety reviews, and practical education for newcomers keep people out of the news for the wrong reasons. Industry leaders can push for cleaner, less hazardous alternatives, and they’re already looking hard. That sort of effort backs up every ounce of progress the chemical world builds, one bond at a time.
Why This Chemical Deserves Respect
Most people don’t cross paths with tert-Butyl peroxyacetate, but if your work brings you in contact with it, you can’t treat it like just another warehouse item. The risks aren’t about inconvenience; this material brings genuine hazards. Peroxy compounds such as this one present both fire and explosion risks under the wrong storage conditions. Years around industrial storage make it clear: even one overlooked detail can get people hurt. That’s why a no-nonsense approach matters so much."
The Basics People Can’t Skip
Once tert-Butyl peroxyacetate arrives, no one should just stack it on a shelf or stow it beside cleaning products. This material reacts to heat, shock, and contamination. For starters, keep it cold. Temperatures creep up past 20°C (68°F) and the breakdown risk jumps. Experience says even a power outage becomes a safety concern if cooling fails. Backups like alarmed refrigerators, with records you check daily, aren’t extra—they’re essential.
Talk to any chemical safety officer, and you’ll hear stories about poor ventilation leading to accidents. tert-Butyl peroxyacetate needs air circulation around both the containers and the storage area. Vapors build up, ignition sources sneak in, and disaster doesn’t care about anyone’s excuses. Invest in air exchanges and explosion-proof fixtures. Simple fans and extension cords just don’t cut it.
Handling and Location Choices Matter
Some may try to minimize risks by reducing stock, but safe storage means more than managing quantities. Separate tert-Butyl peroxyacetate from flammable liquids, acids, and anything that might react. Distance gives you time if something goes wrong, and in chemical storage, time saves lives. Metal shelves spark under impact, so non-sparking shelving wins every time. Flooring should resist spills and stand up to cleaning chemicals after an accident. Near misses have happened over small leaks that weren’t controlled quickly.
People underestimate labeling and records, especially when staff changes or shifts pick up speed. Losing track of one container after a delivery can spiral into confusion during a fire drill. Labeled, locked cabinets—with keys controlled by one or two trusted supervisors—keep out hands that shouldn’t be near such materials in the first place.
Safety Gear and Emergency Planning
A supply of goggles, gloves, and face shields won’t stop a fire, but they keep splashes from sending someone to the ER. Real-life labs keep spill kits within reach of the storage room. Practicing response with real water (not just a PowerPoint) gets people’s instincts tuned to the right moves. If a drum leaks or a fridge dies, waiting for the official emergency team isn’t always realistic. Nearby drench showers and eyewash stations mean accidents don’t turn tragic while help is on the way.
Storing chemicals like tert-Butyl peroxyacetate carries weight—regulatory, ethical, and practical. Checklists or digital monitoring don’t replace real experience and accountability. Companies and universities owe it to workers and the neighborhood to take this seriously. If you doubt the importance, ask anyone who’s seen careless storage go wrong. Lessons learned after an incident don’t just cost money—they can change lives forever.
The Risks Behind the Chemistry
tert-Butyl peroxyacetate doesn’t make headlines, but it brings a basket of risks that deserve some attention. Many who spend their careers working with chemicals spot danger long before trouble strikes, usually by the smell, the way a liquid moves, or a label that sends a signal to keep hands steady. I’ve spent enough time in labs and around manufacturing plants to recognize that some materials seem benign until one misses a key detail on a safety sheet.
This compound falls into the group of organic peroxides. They act as initiators in polymerization, sometimes showing up in resin plants or specialty chemical production. The hazard isn’t rare or hidden: this chemical catches fire easily—not just at high heat, but in normal conditions if accident mixes it with the wrong materials. A splash on the floor or into a different waste drum can light up a room, especially where solvents and heat sources gather.
Fire and Explosion: The Real Challenge
tert-Butyl peroxyacetate readily decomposes, and this process speeds up if it heats up. In my own experience, storage habits matter—temperature controls, sturdy ventilation, and strict engineering checks keep things calm. Skip these, and the runaway reaction could turn even a small leak into an explosion. The U.S. Chemical Safety Board has case files showing injuries from fires caused by improper peroxide handling, and the common theme is always overlooked controls or hasty decisions.
Once decomposition kicks off, the result is hot, dense gases with nowhere to go but out. The blast can shatter glass, burn anyone nearby, and leave toxic byproducts in the air. Sometimes, workers assume a diluted solution offers more safety, and that’s not a guarantee. Dilution lowers risk, but with enough peroxide in a big enough batch, danger lingers.
Toxicity and Health Warnings
Even skipping past the fire hazard, direct contact with tert-Butyl peroxyacetate threatens health. Skin blisters, eyes water and sting, and breathing fumes leads quickly to headaches and sore lungs. Long-term exposure remains less studied but chronic irritation turns up every time a team tracks exposures across months. Personal protective equipment—solid gloves, goggles, and firm spill controls—change the outcome from emergency room visit to just another shift.
Why Proper Handling Makes a Difference
Training makes the real difference. In every plant I’ve worked in, the places with routine safety briefings and real drills handle trouble without panic. Workers there learn to spot leaks fast, wash off spills right away, and recognize the sound of vent fans slowing down. Managers don’t cut corners on protective gear or checklists, even on days when things seem “normal.” It always comes down to everyday habits, not the rare event.
Manufacturers label containers well for a reason. Those bright symbols on the drum aren’t there for decoration—without clear markings, it’s too easy for a barrel to end up in the wrong storage locker or tossed with general trash. Industry stats show that labeling mistakes and missing paperwork remain among the most common sources of chemical mishandling.
Reducing the Harm
The best solutions come from strict control—low storage temperatures, separated flammable materials, regular inspections, and equipment built with chemical compatibility in mind. Teams that build a strong workplace culture where anyone feels comfortable bringing up a small concern tend to avoid the worst outcomes. Having experienced the noise and confusion after a minor spill, I’ve seen firsthand how a few minutes of prevention trumps hours of emergency response.