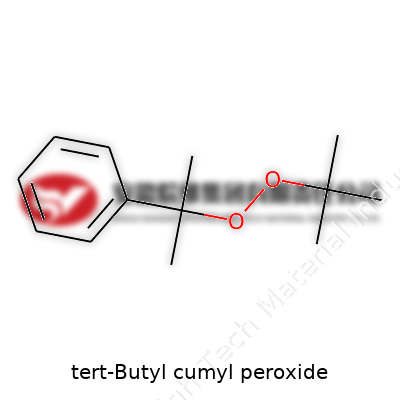
tert-Butyl Cumyl Peroxide: A Deep Dive Into Its Role, Development, and Safety
Historical Development
Chemists have tinkered with peroxides for more than a century, but tert-butyl cumyl peroxide first gained traction in the late twentieth century. Organic peroxides rose to prominence as industry sought more effective ways to initiate polymerization and modify molecular structures for rubbers and plastics. Demand for more stable yet active initiators gave rise to a family of dialkyl peroxides. Among these, tert-butyl cumyl peroxide stood out for a unique combination of thermal stability and reactivity. During the energy crises of the 1970s, synthetic routes became more streamlined and safer, driving down cost and encouraging broader adoption in manufacturing environments across North America, Europe, and East Asia. Many production facilities still use legacy methods from this period, improved for throughput and safety with modern reactors.
Product Overview
Used primarily as a radical initiator, tert-butyl cumyl peroxide serves in the cross-linking of elastomers, the polymerization of styrene and polyethylene, and a handful of specialty coatings. Powder, liquid, and paste forms exist. End users see it as a workhorse compound—one that balances performance with handling requirements. Its shelf life, energy of activation, and compatibility with typical monomers made it the go-to choice for producers seeking reliable throughput in batch and continuous manufacturing alike. Chemical suppliers focus heavily on guaranteeing consistent concentration and purity, aware that even small deviations can disrupt an entire production run.
Physical & Chemical Properties
tert-Butyl cumyl peroxide comes across as a colorless or light yellow liquid at room temperature, carrying a distinct, faint aromatic odor. A molecular weight of 234.33 g/mol, a density close to 0.97 g/cm³, and a boiling point above 200°C give it staying power during high-temperature processes. Decomposition typically kicks in above 140°C, releasing radicals that drive polymerization at speed. It dissolves readily in most organic solvents but resists water, a trait useful in process design. Its peroxide bond lends strong oxidizing potential, demanding a steady hand in storage and use. Labs track temperature, pressure, and exposure to UV to avoid runaway reactions—a lesson hard-won over decades of industrial practice.
Technical Specifications & Labeling
Manufacturers analyze each lot for active oxygen content, ensuring that it falls within tolerances set by global standards. Common purity grades land near 98% active ingredient. Standard labeling underlines hazards: organic peroxide, flammable, and harmful if swallowed, inhaled, or in contact with skin. Labels also carry UN identification numbers and hazard pictograms to comply with GHS (Globally Harmonized System) rules adopted worldwide. Storage advice insists on cool, shaded locations far from incompatible chemicals—especially acids, bases, and metals like copper or iron that catalyze decomposition. Personal experience teaches that tight controls on both inventory and documentation can save a plant from costly recalls or workplace hazards.
Preparation Method
Producer plants synthesize tert-butyl cumyl peroxide most often by reacting cumyl hydroperoxide with tert-butyl alcohol in the presence of acid catalysts, typically sulfuric or p-toluenesulfonic acid. This transesterification normally occurs under moderate heat and vacuum, with byproducts carried away through distillation. The final product might undergo vacuum distillation or chromatography to remove trace impurities. Quality oversight plays a constant role—each batch runs through GC-MS (gas chromatography–mass spectrometry) analysis to confirm purity and rule out high-risk side products. As plant chemists know, overlooking a minor impurity can spoil the initiating power when downstream users need it most.
Chemical Reactions & Modifications
tert-Butyl cumyl peroxide enters the spotlight in radical chemistry. Heat breaks the peroxide bond to generate two radical fragments—a tert-butoxy and a cumyloxy radical. These intermediates attack unsaturated bonds in monomers, driving polymer chain formation in rubber, polyethylene, and styrene. Chemical engineers have manipulated its decomposition temperature and reaction rates by introducing co-catalysts or inhibitors, gaining tighter control over product quality and material properties. Derivatives based on minor substitutions to either the tert-butyl or cumyl groups have led to customized reactivity for tailored applications, such as heat-curing large cross-sections in tire and cable manufacturing.
Synonyms & Product Names
Across the globe, this compound appears under different tags: tert-butylcumyl peroxide, TBKP, 1,1-dimethylethyl(1-methyl-1-phenylethyl)peroxide, and technical product codes from major suppliers. Distributors often use branded names for market differentiation, yet regulatory filings typically refer to the IUPAC or CAS nomenclature. For those procuring materials internationally, cross-checking synonyms avoids misdelivery or incompatible batches—a simple but often forgotten step that means the difference between smooth scaling and production stalling.
Safety & Operational Standards
Any facility using this compound puts safety first, as its oxidative nature and thermal instability lead to real risks during storage, transfer, and use. OSHA and European REACH standards lay out clear protocols: wear chemical-resistant gloves, goggles, and face shields; provide full air exchange or local exhaust in process areas; and segregate storage from combustibles or strong reducing agents. Containers feature pressure-relief capabilities to prevent rupture from gas evolution. Training drills help staff respond if spills or fires break out since, from experience, even small exposures to vapor or liquid can lead to skin irritation, headache, or, more seriously, breathing trouble. Emergency showers, eyewash stations, and clear SOPs keep incidents rare and minor.
Application Area
Factories turn to tert-butyl cumyl peroxide for rubber vulcanization, especially in making tires, belts, hoses, and profiles requiring durable elasticity and chemical resistance. The compound’s radical-forming ability suits the production of cross-linked polyethylene (PEX), a staple piping and cable insulation material. Manufacturers of foamed plastics, adhesives, and specialty resins also draw on its initiator prowess. Certain advanced coatings used in automotive and electronic industries benefit from the precise reaction kinetics this peroxide delivers. For any operation chasing increased strength, longer life, or altered flexibility in polymers, this chemical remains the initiator of choice.
Research & Development
Laboratories across the globe chase ways to improve on the stability and handling of tert-butyl cumyl peroxide. Research teams work on encapsulating peroxides in microbeads or gels to reduce dust and spill risk, seeking safer delivery forms for manufacturing lines. Studies dive into kinetic profiles, looking for ways to lower processing temperatures and energy costs without sacrificing initiator performance. Engineers model thermodynamic properties and reaction networks with computing power unthinkable a decade ago. Industry consortia share best practices across borders, with academic partners narrowing in on green chemistry routes that limit emissions and reduce hazardous waste. Labs use every bit of modern technology—NMR, FTIR, calorimetry—to give factories confidence in each iteration.
Toxicity Research
Scientific work on the health impacts of tert-butyl cumyl peroxide draws on in-vitro studies, animal testing, and historical accident reports. Short-term exposure irritates the skin and mucous membranes. Chronic handling at low levels does not appear to cause cancer, but regulatory agencies urge limiting airborne and dermal exposure as much as possible due to its strong oxidative nature. Accidental ingestion or large-scale releases require immediate medical action to halt respiratory and circulatory complications. Modern toxicology tracks metabolic pathways, breakdown products, and genetic impacts to spot any potential long-term effects ahead of regulatory changes. Medical surveillance in production plants over decades shapes workplace limits and guides new engineering controls.
Future Prospects
The need for higher-performance materials, lighter vehicles, and better insulating properties will keep tert-butyl cumyl peroxide in demand for years. Chemists continue to tweak initiator compounds for lower energy use, easier recycling, and safer handling. Markets in Asia and the Middle East grow rapidly, driving scale and speeding technical innovation. Sustainable chemistry will play a growing role, with greener synthesis approaches set to cut costs and waste. Next-generation applications—like advanced composites for wind turbines or electric vehicle batteries—call for reliable, high-purity initiators. Close collaboration between manufacturers, universities, and regulators will push new standards for purity, safety, and environmental stewardship, making this chemical a fixture of the global industrial toolkit.
A Powerful Tool in Polymer Production
Anyone who has worked with plastics knows the importance of getting the right amount of toughness and durability. Tert-Butyl cumyl peroxide makes this possible. This chemical gets used as an initiator, especially during the process of polymer crosslinking. In straightforward terms, think of it as a spark: the compound helps start or drive chemical reactions that bind molecules together. The result gives us stronger, more flexible plastics—materials found everywhere, from car tires to the wire insulation in your home.
Behind the Scenes with Rubber and Plastics
During my time working in an industrial setting, I watched workers wrestle with the challenge of making materials that could handle daily abuse. Tire manufacturers, for example, rely on this peroxide to toughen rubber. The compound releases energy at a predictable temperature, so you have precise control over the final product. Without this level of reliability, tires would wear out faster, hoses would crack, and medical tubing would lose its flexibility. Products shaped and cured with help from this chemical last longer and cost less to replace.
Cleaner Processing and Long-Term Stability
Factories have always chased ways to improve efficiency. Unlike some older crosslinking agents, tert-Butyl cumyl peroxide works quickly and leaves behind fewer byproducts. That means cleaner conditions in the plant and less waste. Polymers made using this chemical also resist heat and chemicals better over time. Manufacturers don’t spend as much on recalls or warranty claims because their products stand up to real-world stresses. In this sense, this peroxide does more than just move molecules around; it makes modern life more sustainable by reducing the need for constant replacements.
Balancing Safety and Performance
No one wants a workplace accident or a shipment of recalled goods. This peroxide brings a level of safety through reliability, but it demands respect. I’ve seen the lengthy safety briefings and storage protocols at chemical plants. Proper handling and smart engineering controls reduce risks linked to exposure or spills. The industry keeps tight regulations for a reason—any lapse invites trouble. Workers need training, and storage conditions must prevent high temperatures or contamination. Mistakes can cost lives and inflict heavy damage on the surrounding community.
Room for Improvement
There’s growing pressure to lower emissions and reduce the environmental burden of chemical factories. Researchers push for ways to make peroxide-based processes safer and kinder to the earth. That could mean finding smarter packaging, designing quicker clean-up systems, or developing recycling schemes for spent chemicals. Some companies now test alternatives that can match the performance of tert-Butyl cumyl peroxide with fewer environmental impacts, but progress moves slowly. The balance between top performance and safety—both for people and for the environment—still needs attention from both industry and regulators.
Final Thoughts
Tert-Butyl cumyl peroxide works quietly, yet its impact shapes the stuff we use every day. From the car you drive to the medical tools at your local clinic, this chemical keeps things flexible, tough, and reliable. Continued focus on safety, efficiency, and environmental responsibility will steer its use in the right direction, making sure progress doesn’t come at too high a cost.
Understanding the Risks
tert-Butyl cumyl peroxide shows up in many labs as a radical initiator. Its strength lies in its ability to kickstart chemical reactions, especially in plastics and rubber production. But let’s be real, the same qualities that make it powerful can also make it risky. Most folks who’ve spent time in the lab know it can decompose violently if mistreated. That risk puts both people and products on the line.
Personal Safety—Not Just Lab Protocol
Each time I’ve handled peroxides like this one, those safety goggles and gloves weren’t just a nod to protocol. I remember a splash landing on a coat once; the material melted. A warning like that sticks with you. Gloves rated for chemical exposure, goggles with side shields, and a buttoned-up lab coat become everyday armor. Skin absorbs chemicals fast, so even small spills matter. Respirators with organic vapor cartridges provide some breathing room, especially if ventilation lacks punch.
Ventilation and Storage
Working in a fume hood isn’t overkill. This compound releases vapors that you don’t want swirling around. Over time, fumes can trigger headaches or worse. Above all, good airflow keeps vapors away from sparks or open flames. tert-Butyl cumyl peroxide won’t forgive a missed detail, as it can ignite with alarming ease.
Storage makes or breaks safety. I never trusted regular cabinets for peroxide storage—dedicated explosion-proof fridges drop the odds of runaway reactions. At work, I’ve seen labels fade, bottles misplaced, and all it takes is one mix-up to set off a disaster. Store this chemical away from acids, reducing agents, and direct sunlight. A cool, dark space, with clear labeling and secure shelving, cuts down on panic and confusion.
Handling and Disposal
Pouring too much at once, or even scraping residue from old containers, can go wrong fast. Keep quantities small—only what you plan to use. Re-seal bottles right away. If spills happen, don’t use paper towels or rags that could smolder. Peroxide cleanup kits and absorbent materials rated for organic peroxides work better and don’t fuel decomposition. Dispose of all waste through hazardous waste channels. Pouring leftover peroxide in the sink isn’t just lazy; it flirts with environmental trouble and regulatory fines.
Training and Emergency Preparedness
I’ve seen seasoned chemists freeze up when alarms go off. Practice counts. Safety training shouldn’t only cover basic spill response; everyone in the space, from interns to old hands, benefits from regular drills. Know where the eyewash and safety shower stand. Fire extinguishers for Class B and C fires belong close, not buried under clutter. Find out if a spill containment kit suits peroxides, since not all do.
The Bigger Picture
Some companies now tie bonuses to lab safety records. Beyond rules and reminders, that approach boosts accountability; people look out for each other because they have a personal stake. Tighter internal policies combined with practical training shrink risk for everyone. tert-Butyl cumyl peroxide brings power, but never without responsibility. Respecting its hazards, using the right protective gear, storing it smartly, and knowing what to do in a pinch all matter. In my lab days, sweating the details kept people safe again and again. That’s the path that never gets old.
Getting to Know tert-Butyl Cumyl Peroxide
Many people outside labs might never stumble on the name tert-butyl cumyl peroxide. This compound matters for anyone working with plastics and rubbers. The formula, C17H26O2, can look like a simple string of letters and numbers. Still, those symbols tie directly to the stuff that shapes products we all touch daily—shoe soles, car interiors, even golf balls.
What Does This Formula Show?
Twenty-six hydrogens, seventeen carbons, two oxygens. In practical terms, the spacing of the carbon atoms makes this molecule bulky, and the peroxide group means it can easily split apart to release energy. Good chemists know that this ability to break down makes tert-butyl cumyl peroxide valuable for starting other chemical reactions. Factories want it when they need to kick off polymerization—the process that turns liquid plastic precursors into solid forms.
I’ve wandered through industrial labs filled with odd-smelling vapors and endless warnings about safety. You notice the peroxide bottles kept far away from anything flammable, and you smell the odd hint of sharpness that lingers in the air even with good ventilation. The workers treat these chemicals with respect for a good reason. Peroxides like this one act as radical initiators—they start chain reactions, which, without good oversight, can escalate quickly and dangerously.
Why Should Ordinary People Care?
The end consumer rarely thinks about the chemistry in a sneaker sole, but that chemistry makes a real difference. tert-Butyl cumyl peroxide’s formula and structure land it squarely in the family of heat-activated agents. This means it works at high temperatures—the same kinds found in heavy-duty plastic molding equipment. Using the wrong initiator can lead to slower manufacturing or weaker products.
Knowing the formula also connects to broader topics like environmental safety. This molecule breaks apart in a predictable way, leaving byproducts that regulators watch. Understanding the strengths of that oxygen-oxygen bond, and how it splits, can help engineers design processes that produce less pollution. Just as importantly, the correct calculations based on that formula keep workers safe.
Safety and Handling: Lessons Learned on the Job
Early in my lab career, a more experienced chemist caught me mishandling a sample of a related peroxide. He didn’t scold; he pulled out the material safety data sheet and showed me the breakdown products—what gases we’d inhale, what residue we’d handle. That experience stuck. The structure revealed by the chemical formula dictates not just how a substance reacts in a controlled flask, but also how it behaves if something goes off-script. tert-Butyl cumyl peroxide needs cool, stable storage and careful measurement. Its formula hints at risk and reward, all depending on how humans interact with it.
Solutions Moving Forward
People in industry can take small steps to help everyone—including themselves. Always double-check labels and formulas. Push for modern storage and containment. Work toward greener, less hazardous alternatives wherever possible. Encourage open dialogue about chemical risks, especially among newer team members. These habits, all rooted in respect for a molecule’s formula, can mean the difference between safe processing and costly accidents. The formula of tert-butyl cumyl peroxide matters far beyond the textbook—it's woven into the strategies that keep products strong and people healthy.
Understanding the Peroxide
tert-Butyl cumyl peroxide doesn’t show up in everyday life, but for people in plastics and rubber plants, the name brings a little tension into the warehouse. Peroxides like this push chemical reactions hard, which makes them useful for manufacturing. They also bring risks, much bigger than most raw materials. Run-ins with peroxides don’t go well—just ask anyone who’s handled leaky containers or forgotten about temperature controls for even a weekend.
Sensitivity and Stability: A Fine Balance
This peroxide does useful work because it falls apart easily, churning out radicals that drive polymerization. That same instability means a poor storage plan quickly turns dangerous. Think heat, sunlight, or metal contamination—a small error leads to fire, even explosion. Most plant workers know at least one story where improper storage caused a scare or chemical loss. Responsible companies make those stories rare by nailing down storage methods and following them every day.
Temperature Control Makes All the Difference
Heat spells trouble. A short rise above the safe zone accelerates decomposition, spoiling the raw material and risking uncontrolled reactions. Cold rooms or dedicated explosion-proof fridges are standard. Most keep these peroxides at 2–8°C, far from any boiler room or sunlit loading bay. At one site, a failed cooling system forced an emergency inventory review—workers needed to double-check every bottle and send compromised stock for safe disposal. People trust monitoring equipment with alarms to catch failures early. New sensors keep logs, so managers spot trends before they become headlines.
No Room for Guesswork with Containment
Chemical compatibility stays front of mind. Peroxides attack metals, and organics like cardboard or oils don’t last long nearby either. Storage often involves original containers with special liners, kept on spill trays that funnel leaks away. Pallets never go directly on concrete or near oxidizable substances. In overcrowded warehouses, one oversight can cause a minor leak to escalate, so clear labeling and designated shelving help everyone check containers quickly—especially during audits.
Segregation Prevents Chaos
Peroxides can’t share space. They react with acids, bases, or just about anything with an active hydrogen. Warehouse layouts always separate these chemicals from incompatible substances, and well-run plants use barrier walls or separate rooms. Labels go beyond regulatory requirements—oversized, color-coded markers signal risk at a glance. Even janitorial staff, who might not work with peroxides directly, benefit from clear signs that keep cleaning equipment and rags at a safe distance.
Training Anchors Everything
Protocols mean little without real-world buy-in. Teams revisit emergency plans and containment steps every few months. Story-sharing about prior mistakes, anonymous near-miss reporting, and regular refresher training all make a difference. One site manager explained how turning “checklists” into “walkthroughs” increased participation—new hires spot broken seals or outdated stock before problems get buried. Training also covers protective gear. Goggles and nitrile gloves aren’t optional; seasoned workers keep gear within easy reach, no matter how familiar they become with the routine.
Building a Safe Routine
Experience and vigilance hold the line. From logbooks to labeling, from cooling alarms to periodic drills, people create day-to-day safety around tert-Butyl cumyl peroxide by sticking to proven systems. Success comes from small daily decisions—quick checks, honest reporting, and a respect for chemistry’s unpredictability. Proper storage isn’t about box-ticking; it’s the sum of careful habits that protect people and investments, year after year.
Why This Chemical Needs Careful Handling
You don’t have to be a chemist to care about chemicals like tert-Butyl cumyl peroxide. It shows up in manufacturing, research, and even some industrial workplaces. This stuff does real work as a catalyst in polymer production and curing resins. That usefulness carries a load of risk, not just for people who mix and move it, but also for those nearby. From firsthand experience working alongside industrial safety teams, I’ve learned you can’t cut corners around compounds like this one.
What Makes tert-Butyl cumyl peroxide Hazardous?
Most folks see white crystals or a slosh of liquid and don’t think twice. But this chemical packs a punch long before anything gets spilled. It’s an organic peroxide, so it breaks down and releases energy fast—sometimes explosively—if it gets too warm or is knocked around. Safety data spells this out: once it overheats, it can burst into flame without warning. That’s not just a mess. It triggers fires that most fire departments respect, using only special foam or dry chemical agents, never water, to tamp them down.
It also gives off fumes that can sting your nose and eyes in seconds. Standing close by, I’ve seen folks cough and tear up just from unsealing a drum in a drafty shop. If enough vapor gets into the air, it can make you dizzy or worse. Longer exposure eats at health quietly—affecting the respiratory tract, aggravating asthma, and possibly attacking the central nervous system with regular exposure.
Long-Term Environmental Impact
After an incident at a plastics plant, we had to chase down a spill of this stuff more than once. Here’s the thing: tert-Butyl cumyl peroxide doesn’t just vanish. It seeps into drains, messes with the water, and brings the risk of contamination to soil and ground water. Fish and aquatic life take a beating from even small amounts. The compound breaks down, but not quickly or cleanly, adding more variables to waste handling and disposal.
Exposure Prevention: What Real Workplace Experience Teaches
It’s easy to wave a binder of safety sheets and call it training, but people listen up with real-world examples. One worker took off gloves just a minute too soon and burned his skin. Once, a shipment warmed up in an unventilated truck, and drivers noticed a sharp smell and got headaches within minutes. Everything points to one solution—constant vigilance. Storage below 30°C, limited light, and keeping sealed containers isolated works best. Gear matters, too. Gloves, goggles, and aprons are a must, not just a suggestion.
Emergency plans save more than compliance—automatic sprinklers using the right suppressant, good local exhaust, and fast access to safety showers stop small mistakes from turning into major disasters. I’ve seen shop teams run drills blindly, but it’s the hands-on training and clear communication that made the difference.
Stronger Oversight and Personal Responsibility
Regulation can nudge companies in the right direction—OSHA and EPA rules exist for a reason. But company culture moves the needle. I’ve watched seasoned operators quiz new hires on signs of peroxide decomposition and rehearse responses to vapor alarms. Everyone from the shipping clerk to the plant manager needs to know the signs of danger and not brush them aside.
Working with tert-Butyl cumyl peroxide demands constant respect. It serves industry well, but demands nothing less than serious care every step of the way.