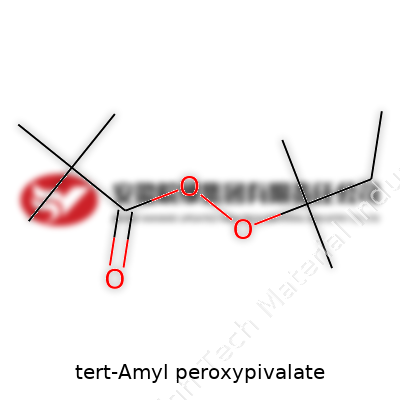
Tert-Amyl Peroxypivalate: Exploring Its Role in Modern Chemistry
Historical Development
Organic peroxides began to shape the chemical industry during the early twentieth century, yet tert-Amyl peroxypivalate never drew the spotlight until manufacturers sought out new, safer initiators for polymerization. Early research centered around methyl ethyl ketone peroxide, but safety concerns kept people on their toes. As industry scaled up, chemists and engineers watched accidents and set their sights on alternatives. Tert-Amyl peroxypivalate offered a middle path. Its structure delivered the right punch over other organic peroxides, so labs and plants gradually adopted it, tweaking process conditions to fit its reactivity profile. Observing industry adoption over years, people recognize that those few safety-driven changes in initiator design didn’t just protect workers; they made high-value polymers possible at commercial scale.
Product Overview
Tert-Amyl peroxypivalate finds its main calling as a radical initiator, especially during the manufacture of PVC and acrylics. This colorless to pale yellow liquid doesn’t just exist in the world of technical datasheets—in many plants, it’s a cornerstone of emulsion and suspension polymerization. Companies favor it not for trendy reasons but because its properties bring real utility. Suppliers deliver it stabilized and package it according to strict regulations to protect users from violent decomposition. Based on my own work in industrial production, what stands out about this peroxide ties to its reliability in starting reactions at low to moderate temperatures, which saves energy and reduces risks associated with elevated pressures.
Physical & Chemical Properties
The technical properties of tert-Amyl peroxypivalate shape how it can be moved, stored, and used. Its boiling point sits lower than water, and it breaks down on its own even below room temperature without stabilizers. Liquid density hangs around 0.95 g/mL, and solubility trends favor organic solvents over water. Handling it in the lab or on the plant floor feels a bit intimidating: it can explode if you even think of mishandling it, especially when dry or isolated from its carrier. The compound contains a peroxy bond that splits easily, releasing radicals that spark chain reactions in manufacturing. There’s an unmistakable acrid odor when vapor escapes, a warning sign not to ignore. Technicians develop a healthy respect for these signs; it keeps accidents from happening.
Technical Specifications & Labeling
Every shipment of tert-Amyl peroxypivalate comes marked with hazard diamonds and strict documentation in line with national and international transport codes. Labels go beyond legal compliance—they warn real people about explosion hazards and poisoning risk. I’ve seen packaging double-lined and shipped in refrigerants, capped with vented stoppers, with paperwork listing product purity, stabilizer type, assay values, and recommended shelf lives. MSDS entries describe both molecular details and practical emergency actions, such as how to quench decomposing material or evacuate a leaking container. Working with regulatory auditors, companies pay special attention to labeling, aware that even a simple misprint can trigger a stoppage or a recall.
Preparation Method
Crafting tert-Amyl peroxypivalate starts with a controlled reaction between tert-amyl alcohol and pivaloyl chloride in the presence of hydrogen peroxide. The process runs under low temperatures, moderated by careful addition of acid scavengers and stabilizers. Chemists watch reaction conditions closely—temperature swings or contamination invite violent reactions. Production lines use jacketed reactors, chilled brine loops, and remote monitoring to guide the process safely from batch to batch. Purification relies on washing away acid and unreacted starting material, then stabilizing the end product with phlegmatizers before packaging. Each step aims for a clean, safe peroxide, learning from past lab mishaps where shortcuts ended in disaster.
Chemical Reactions & Modifications
Tert-Amyl peroxypivalate earns its place in radical chemistry by splitting at low activation energies to produce carbon-centered radicals. In polymerization, the generated radicals start new chains from vinyl monomers—acrylics, styrenics, and especially PVC. People exploring new products try blending or modifying the peroxide with synergists and co-initiators, hoping to nudge yields or control molecular weights. In research and on the factory floor, technicians sometimes shift dosing or temperature profiles to fine-tune properties in the final polymer, understanding that small changes in handling can have outsized effects. In side projects, chemists explore modifications to reduce volatility or improve compatibility with tricky monomer mixtures, yet classic tert-Amyl peroxypivalate rarely gets sidelined because it works so well.
Synonyms & Product Names
Tert-Amyl peroxypivalate pops up in sourcing under various names. Companies catalog it as TAPP, tert-pentyl peroxypivalate, or use proprietary trade names depending on region or supplier. These synonyms matter in real purchasing and audit scenarios. I’ve witnessed confusion in both procurement and hazard training when mixtures or branded products get swapped, as each name ties to a specific stabilizer or dilution. Clear communication between vendors and end-users keeps workplace safety high and avoids regulatory missteps.
Safety & Operational Standards
Every person handling tert-Amyl peroxypivalate wears respect for its hazards right on their sleeve. Regulatory guides call out temperature limits for storage, often below 10°C to slow down decomposition. Ventilated storage, fireproof segregation, and ready access to spill containment gear get drilled into operating procedures. Training goes beyond paper drills—technicians run what-if scenarios and rehearse emergency shutdowns, because memory and muscle save lives when alarms sound. Disposal routes funnel into chemical deactivation, often through reduction to less risky compounds before incineration. Risk managers keep detailed logs and track near-misses; companies can’t afford to take shortcuts with a compound that could detonate or poison a room with a small mistake. Insurance firms and inspectors take special interest in any site where organic peroxides move in bulk.
Application Area
Most people only encounter the results, never the raw chemical. Plastics—flexible, rigid, colorful—owe their structure to radical initiators like tert-Amyl peroxypivalate. Production of PVC pipes, wall panels, coated fabrics, and certain adhesives flow through processes sparked by this compound. Offshore, operators in oil and gas sometimes reach for it to tweak resin chemistries in specialty coatings and sealants. Researchers hunting for biodegradable plastics, anti-fouling paints, or even medical-grade polymers sometimes run side experiments to see if TAPP-based initiators bring better control or lower toxicity byproducts compared to older, harder-to-handle peroxides. Despite its hazards, the reliability and established processing protocols make it a preferred ingredient for large-volume manufacturing where minor cost changes carry big bottom-line impacts.
Research & Development
Academic and industrial scientists keep tinkering, trying to stretch the boundaries of what tert-Amyl peroxypivalate can achieve. Polymer chemists, in particular, push it through controlled radical processes to dial-in specific chain architectures or functionality. Some teams partner with green chemistry groups, looking for ways to recover spent initiators or convert waste into safer forms. Equipment manufacturers, meanwhile, tweak reactor and containment designs to offer safer large-scale options, using lessons from near-misses and published incident reports. Open access journals now capture side-by-side studies of different peroxides, giving buyers more data to weigh reactivity, cost, and safety. Research on its influence in emulsion versus bulk processes sometimes yields incremental gains, which add up across plants running thousands of tons per year. From my experience reviewing industry conference proceedings, most new advances trickle into technical guidelines and plant standard operating procedures over a span of years, not months.
Toxicity Research
Tert-Amyl peroxypivalate has prompted scientists and health professionals to dig deep into its toxicology. Toxic effects show up quickly in poorly ventilated labs—skin burns, eye irritation, and respiratory distress don’t wait long to announce themselves. Chronic exposure studies signal risks of organ damage and even mutagenicity, though its main exposure window lands in occupational use. Real-world incidents reinforce the lessons: a single spill can drive an evacuation, and delayed reporting inflates hazard. Labs track threshold limit values and monitor for metabolites in wastewater streams. Ongoing concern lands on breakdown products, since incomplete destruction of the peroxy bond during incineration can leave sneaky organic acids behind. Workers’ health teams check personnel regularly, and push for continuous upgrades in handling protocols as fresh research uncovers new risks or better antidotes.
Future Prospects
Looking down the road, tert-Amyl peroxypivalate faces mounting pressure from both inside and outside the chemical industry. On one side, market demand for durable, cost-effective plastics keeps production humming. On the other side, regulatory agencies around the world eye peroxides more closely, imposing stricter storage, transport, and emissions controls in response to higher-profile accidents and calls for greener chemistry. Companies investing in automation, sensors, and new formulations hope to shave down hazards while boosting productivity. Researchers continue their hunt for safer alternatives that match performance without legacy risks. Those who work with TAPP will keep balancing innovation against caution, updating technical controls and investing in training for the next generation of chemists and process operators. If future improvements in catalyst design or recycling tech open doors to new uses or greener management, the lessons of the past—earned through care, rigor, and tough experience—will continue to shape how the industry moves forward.
The Chemistry Behind Progress
Tert-amyl peroxypivalate belongs in the toolbox of chemical producers. Its main use connects to polymer chemistry. Most people walking through their daily lives rarely think about how plastics or coatings come together. Chemical initiators like this one make the difference between a sticky mess and a finished product with the right strength and appearance. That detail might sound technical, but it touches a lot more of our world than most folks expect.
Where Manufacturers Rely on It
This compound finds its main job as a radical initiator. Picture starting a chain reaction that builds huge polymer chains. Acrylic resins, especially those you find in paints or adhesives, often need reliable starters to get the process going. Tert-amyl peroxypivalate steps in because it breaks down in a controlled way once heated—releasing radicals that drive polymerization. That precise control isn’t just a bonus. Factories betting money and safety on each batch value predictable reactions. Fast, reliable, and stable—these qualities have earned it a place in the production line.
Impact on Everyday Products
Some skeptics wonder why any of this matters if people can’t pronounce the names. If you care about the durability of road markings, the finish on your car, or the clarity of inkjet prints, you care about what goes into the production. Not every initiator delivers the balance between power and predictability. Some react too quickly and burn through raw materials. Others lag behind and slow the process. This specific compound gets chosen when temperature matters—a lot of plant managers like how it opens up scheduling flexibility in warmer seasons, trimming energy costs and waste at the same time.
Risks and Handling Concerns
Peroxides don’t offer their benefits for free. Most folks who have worked around this kind of chemistry get trained to respect what it can do. Improper storage or mishandling turns these blends from a helpful tool into a hazard. Fires and explosions are not unknown in facilities that cut corners. Chemical companies and manufacturing plants carry the burden of training, safety audits, and regulatory paperwork for a reason. That vigilance came from years of hard lessons, where a single unnoticed spark or warm room ruined supplies and injured workers.
Keeping Safety at the Forefront
Clear labeling, temperature control, and personal protective equipment stand out as practical measures. I talked to a friend who manages a coatings factory; their crew treats initiators with the same respect given to heavy equipment or acids. Digital monitors in storerooms track ambient temperature, ensuring the product stays within a safe zone. Mistakes in this arena make national news for a reason. The Open Chemistry Council and similar watchdogs maintain public lists of recommended protocols, pushing for constant training updates and smarter storage solutions.
Looking for Better Alternatives
Some companies keep hunting for alternatives, hoping to replace peroxides with milder or more environmentally friendly options. But replacing a well-understood workhorse isn’t easy. Success depends on balancing performance with health and environmental impact. Researchers study green chemistry routes, like enzyme-based polymerizations or UV-triggered initiators. No breakthrough caught on at scale yet, but the push for safer, greener processing stays strong, often driven by demands from both regulators and consumers.
Building Trust Through Transparency
Public trust in chemical manufacturing never comes easily. Companies that use tert-amyl peroxypivalate publish safety data sheets, join industry quality programs, and participate in environmental reporting. That openness helps not just workers, but also neighbors living near production facilities, stay informed. Real understanding grows when technical information gets explained in plain talk—safety, performance, and progress going hand-in-hand. As someone who’s followed these developments, I see steady improvement, but no reason for complacency.
Understanding the Ingredient
tert-Amyl peroxypivalate serves a key role in the chemical and plastics industries as a polymerization initiator. The compound’s strong oxidizing ability comes with its share of risks. I’ve seen even experienced lab staff lose their calm around organic peroxides—there’s a reason for that.
The Truth about Hazards
You want to know what makes tert-amyl peroxypivalate so tricky? It doesn’t play around when it comes to heat or physical shock. Exposure to a warm room or a careless bump can spell trouble—meaning fire or explosion. Several safety data sheets warn about decomposition above 30°C. Now, that’s lower than the temperature inside a parked car on a summer day. For storage, coolers or temperature-controlled vaults take priority. This isn’t a “back of the warehouse” candidate.
Peroxides also release harmful vapors. Accidental inhalation or skin contact can cause irritation and injuries. I’ve spoken to chemists who swear by thick gloves, goggles, and even face shields. Opening a bottle in open air, far from ignition sources, pays off.
Strict Temperature Control
With organic peroxides, chillers do the heavy lifting. tert-Amyl peroxypivalate prefers a stable, low temperature, typically below 20°C. Unstable storage can speed up decay and push vapor pressure higher. Most labs use explosion-proof refrigerators with clear signage. Regular spot checks and alarms don’t get skipped. Manufacturers often supply the material in insulated shippers and stress immediate transfer on arrival.
Choosing the Right Containers
This is no time for careless choices. tert-Amyl peroxypivalate comes in HDPE drums or specialty glass, far from sources of contamination. Metals like copper or iron turn small leaks into major incidents, so storage away from metals matters. Containers always feature extra seals—these peroxides seep fumes if left unchecked.
Separation from Incompatibles
I’ve seen the results of missed shelving rules: two incompatible chemicals seat inches apart, risking cross-contamination. Strong acids, bases, reducing agents, or even sunlight—each turns this peroxide dangerous. Warehouses keep peroxides in separate, ventilated lockers. Regulations in Europe and the US echo this, setting minimum “isolation” distances.
Why Routine Training Counts
Safety culture can’t just sit on paper. Regular, realistic training sessions help staff spot spills, leaks, or container swelling before events grow unmanageable. Emergency eyewash and shower stations line the corridors close by. Spill kits stocked with inert absorbents and neutralizing agents should stay within arm’s reach. Every new worker gets a demo, not just a checklist.
Disposal Lessons Learned
Old, expired tert-amyl peroxypivalate doesn’t get dumped down the sink. Accumulated peroxides demand expert disposal—incineration, not landfilling or sewer systems. Licensed hazardous waste contractors offer pick-up and documentation. Mishandling disposal not only puts people in danger, but also draws fines from regulators.
Best Practices Keep People Safe
Careful storage, precise temperature control, and separation keep operations smooth and accidents rare. Investing in proper training and reliable containment might slow things down up front, but every chemist I know values their safety above speed. In this business, caution isn’t extra—it’s essential.
Real Hazards Behind the Formula
tert-Amyl peroxypivalate doesn’t get much mainstream attention, but anyone in a lab or industrial setting knows it holds a reputation for being a tricky chemical. I remember my first exposure to organoperoxides came with a safety briefing that left even the seasoned chemists paying close attention. This compound falls into the family of organic peroxides known for kickstarting polymerization reactions, especially for acrylics. The trouble starts with its instability—peroxides break down easily, and when they do, you can expect heat, gas, or even explosive decomposition. That means, next to a workbench or packed in transit, an innocent bottle of this stuff can suddenly turn into a big problem.
The health effects stick in my mind just as much. Even a splash on skin leads to burns or strong irritation. Breathing in its vapor isn’t just annoying—prolonged exposure can give you headaches, nausea, or much worse depending on concentration. Direct contact with eyes spells trouble, possibly causing permanent damage. Doctors and safety officers always stress not to take that lightly.
Fire and Explosion Dangers
Fire risk climbs higher because tert-Amyl peroxypivalate doesn’t just catch flame; it helps other things burn. Once temperatures rise, the chemical can run away with itself—thermal decomposition is a very real fear. In my experience, even a slight temperature slip in storage sparks alarms, which makes sense. Accidental mixing with traces of metal powders or acids can push things from dangerous to catastrophic, so keeping records on everything nearby is standard practice.
Practical Safety Steps Everyone Should Follow
Solid safety steps stem from experience, regulations, and being able to picture what could go wrong. Working with this peroxide, I always double up on personal protective wear: face shield, goggles, gloves that resist solvents, and lab coats. Don’t settle for a simple fume hood—every procedure calls for local ventilation and, if possible, explosion-proof electrical setups. In the lab where I worked, there was never any food, drinks, or casual containers anywhere near organic peroxides; contamination risks simply stack up otherwise.
Storage guidelines amount to a sort of unwritten rulebook among chemists. The bottles stay in special temperature-controlled cabinets, far from sunlight, heat sources, or acids. Anything above 20 °C makes things dicey, so most places use alarms and regular thermometer checks. Sudden pressure changes or bumps cause significant stress, so moving these chemicals becomes a careful, planned action.
For spills—nobody wants to think of that, but I learned from a classmate’s mistake that speed trumps everything. Spills get covered with inert material, swept up, and sealed in fire-resistant waste containers. Fire responders need to know the exact chemical, because standard extinguishers do little if decomposition has built up pressure or toxic fumes. Having a spill kit and fire blanket within arm’s reach can make all the difference.
Solutions That Work in Real Labs
Engineering controls form most of the backbone—isolated storage, automatic temperature alarms, and limited quantities outside secured cabinets. Every safety briefing, drill, or review grows from prior near-misses or hard-learned lessons. I’ve seen regular chemical audits help spot issues, flag expired stock, or find cross-contamination before it matters.
With new workers, in-depth training always beats speed on the job. Detailed labels, routine monitoring, and keeping water and incompatible agents far away move risk from personal worry to shared responsibility. Testing incident response and updating protocols, not just copying-pasting rules, lets teams adapt as their knowledge grows.
tert-Amyl peroxypivalate challenges facility managers and chemists to treat every gram with respect. Every safety step, from PPE to storage, reflects lives and property protected by those who know the risks need attention—every single day.
Understanding the Clock on Chemical Shelf Life
Every lab tech and production specialist runs into the same question from time to time: how long will this bottle of tert-Amyl peroxypivalate last before it turns into a hazard or loses its punch? I remember the nervous look a new colleague gave me the first time they found a slightly dusty drum with a date three summers past. This isn’t just about checking a box on a safety audit. It’s about avoiding accidents and lost productivity, and sometimes, the difference between making deadline or causing downtime.
The Science Behind It
tert-Amyl peroxypivalate acts as an initiator in polymerization processes. This chemical carries a peroxide group, which breaks down over time. Heat, sunlight, and even its own gradual decomposition chip away at its reliability. Most manufacturers mark the shelf life at six months up to a year if stored at recommended temperatures, typically below freezing. My experience in the field tells me to watch not just the date, but the storage log. That one time the cold room lost power for an afternoon in July, we had to reassess everything on those shelves. Good lot tracking paid off.
Risks That Don’t Fade With Time
Some folks think an old bottle is just less effective. Unfortunately, spoiled peroxides do more than lose energy. They can form unstable byproducts, which boost the risk of a runaway reaction or, worst case, an explosion. The National Fire Protection Association and manufacturer bulletins spell it out: expired peroxides don’t just fizzle, they threaten people and property. Incidents in university labs and industrial plants have highlighted those dangers, often traced back to old, improperly stored material.
Human Error and Best Practices
Shelf life isn’t just a theoretical limit. It reflects what happens when real people interact with chemicals daily. Temperature logs, proper labeling, and first-in-first-out stock management reduce risk. I’ve worked in places where fear of waste led some staff to press on with “probably fine” stock. Each batch I’ve seen re-tested after a best-before date shows unpredictable results—not just weak initiator but sometimes way off-gassing and discoloration, obvious flags. The cost of retesting and safely disposing of expired chemicals often outweighs any savings from stretching a product’s use.
Solutions For Safer Handling
Digital inventory tools help keep teams honest. I’ve set notifications on my phone to check up on flagged lots before the deadline creeps up. Simple steps like using color-tagged labels, isolating temperature-sensitive materials, and running regular chemical safety training prove more effective than any shelf-life extension technique. Suppliers can sometimes offer stabilizers or improved packaging, but no trick substitutes for cold, dark storage and vigilance.
Takeaway From the Lab Floor
Shelf life matters because chemicals like tert-Amyl peroxypivalate aren’t forgiving of mistakes. Missing a replacement order or getting lazy with temperature checks puts people and projects on the line. In every setting I’ve worked, the teams that put safety ahead of squeezing out every last drop stay productive and, just as important, go home in one piece.
Understanding the Risks of tert-Amyl Peroxypivalate
tert-Amyl peroxypivalate stands out among industrial chemicals because it poses real hazards. This compound breaks down quickly with heat, light, or rough handling. It’s a strong oxidizer and decomposes to release gases, pressure, and toxic fumes. The real danger shows up when folks try to move, open, or toss out leftover material without solid preparation. This isn’t the type of stuff anyone wants seeping into local landfills or washing down a drain. Just a little bit catching the wrong spark puts people and property at risk.
Why Old Habits Don’t Cut It
For years, some people stashed or diluted problematic chemicals, hoping risk would disappear. That approach never held up. News stories have warned about painful burns and accidents linked to improper handling of peroxides like this. Landfills never break down these peroxides safely. Pouring chemicals down the sink leads straight to surface water, poisoning aquatic life and sometimes disrupting sewer treatment processes. Fines stack up fast for people or companies who cut corners. Taking the right steps avoids bigger legal and health messes later.
Safer Disposal: What the Pros Actually Do
Teams trained in chemical safety start with containment. tert-Amyl peroxypivalate holds up in tightly sealed, completely dry, shaded containers—usually the exact original packaging. Handling usually goes down in cool spaces, using non-sparking tools. Nobody opens a peroxypivalate drum near smoking, flames, or heat sources.
Workplaces with waste use the services of certified hazardous waste companies. These professionals don’t just haul it away. They neutralize, stabilize, or fully decompose peroxypivalate—sometimes by controlled incineration in specialized facilities where heat and filtering technology turn dangerous leftovers into manageable vapors and water, without surprise fires. Every batch gets documented, with tracking numbers and signatures, so there’s a clear trail from origin to disposal. Any spill, no matter how small, usually gets scooped up with absorbent pads and locked into waste drums labeled by type and hazard, never passed off with regular trash.
Legal Ground Rules Everyone Follows
US law and equivalents in Europe and Asia label this stuff as hazardous waste. The Resource Conservation and Recovery Act (RCRA) in the States, for example, spells out how peroxides must be treated. Unmarked barrels, leaks, or slipups mean costly shutdowns and sometimes end up on the nightly news. Regulators ask for proof of staff training and documented methods for handling and disposal. The same rules reach all the way down to university labs, where I once handled quarterly peroxide sweeps—triple-checking expiration dates and signing each removal log. Sometimes the difference between a regular day and one that requires the fire department boils down to these small habits.
Room for Improvement and Smarter Habits
Smaller companies and research groups sometimes lack dedicated safety professionals. Local fire authorities and university extension programs offer free classes on hazardous waste and can even recommend trusted disposal services, helping plug this gap. Digital logs and automated labeling systems now keep track of who opened, moved, or disposed of every chemical. Consumer awareness, clear signage, and honest conversations about hazardous waste matter as much as new regulations. Even if the job feels like an endless paperwork loop, consistent documentation and up-to-date training save lives and the bottom line in the long run.