N,N-Dimethyl-4-Nitroaniline: Context, Chemistry, and Its Future
Historical Development
Synthetic dyes began transforming industries in the late 1800s, and N,N-dimethyl-4-nitroaniline surfaced as a critical intermediate just as chemical factories switched from natural to synthetic colors. Its strong yellow hue grew valuable as European companies expanded dye production. Early patents from Germany, France, and Britain reveal its place in both research and the commercial sphere. Looking at this progression, I see how competition drove both innovation and stricter controls after toxic incidents in crowded factories. The material’s long record reminds us that new discoveries often arise not in isolation, but as byproducts of curiosity, practicality, and sometimes mistakes.
Product Overview
This molecule stands out because of its dual roles: acting as a colorant itself and sparking further reactions. What captures chemists’ attention is the balance struck between the electron-donating dimethylamino group and the strongly withdrawing nitro group. In yellow pigments, small changes here can sharply shift hue or performance during processes. Broadly, you'll find it in dyes for textiles and inks, as well as a reagent for research purposes. It’s not a huge-volume commodity, but in specialty chemistry, its importance doesn’t shrink. Companies who rely on custom colors and advanced intermediates continually source it for custom syntheses.
Physical & Chemical Properties
Usually encountered as a crystalline yellow solid, N,N-dimethyl-4-nitroaniline carries a melting point in the 74-76°C range. Water won’t dissolve it easily, yet common organic solvents like ethanol and acetone make it usable for lab and manufacturing work. Its structural formula—C8H10N2O2—reflects a benzene ring carrying those two key functional groups. This setup gives the material strong optical absorption, which shows up in its use as a dye, and chemical resilience. One thing I’ve noticed is its smell—a slight sharp, nitro-aromatic odor signals its presence in small-scale workspaces. Stability at room temperature reduces handling headaches, but exposure to light or strong acids starts breaking it down and can stain everything yellow.
Technical Specifications & Labeling
Different applications demand different levels of purity. In textiles or ink manufacturing, 98% pure usually suffices. Analytical applications push for purities above 99%. Some suppliers offer it as a fine powder or compacted granules, each format useful depending on the feed system. Each bottle or bag carries standard hazard labels to warn about skin, eye, and respiratory risks, along with batch data and shelf-life details. Consistent labeling helps trace each sample through research or production lines, and finished lots often include a certificate summarizing purity, mass spectrometric info, and absence of problematic byproducts, matching international standards.
Preparation Method
Traditional preparation methods usually start with p-nitroaniline or its derivatives. Through methylation, most often via reaction with dimethyl sulfate or iodomethane in a basic medium, the dimethylamino group gets installed. This particular step demands careful temperature control, use of fume hoods, and reliable waste disposal since reagents and byproducts pose health risks. In academic labs, smaller-scale preparations utilize milder reagents and greener approaches—occasionally swapping out methylation agents for less hazardous ones. The chemical engineering side deals with scaling up reactors safely and preventing runaway reactions, especially important because poorly managed exotherms can lead to accidents, as shown in a few industry case studies over the years.
Chemical Reactions & Modifications
Because the molecule boasts both an electron-rich and an electron-poor side, it opens up diverse routes for organic chemists. Reductive conditions target the nitro group, pushing it to an amine, which becomes an entry point to more complicated azo dyes or pharmaceuticals. Electrophilic aromatic substitutions influence ring positions based on the substituent effects already present. Alkylations, acylations, and nucleophilic aromatic substitutions all play out depending on process needs. I’ve seen it pop up in undergraduate labs where its color change helps teach principles of spectroscopy or as a substrate for kinetics experiments. The ease of functional modification makes this a steady feature in research, not just industry.
Synonyms & Product Names
This is one chemical where the synonyms can get long and confusing: 4-Nitro-N,N-dimethylaniline, p-nitro-N,N-dimethylaniline, C.I. 10375, and sometimes “dimethyl yellow base” in the dye industry. Catalogs and regulatory databases carry all these synonyms to help users track it globally. Larger chemical suppliers assign additional product codes for internal tracking. On many bottles, you might spot international harmonized codes and hazard pictograms following both European and American classification requirements.
Safety & Operational Standards
Toxic effects, especially when inhaled or absorbed through skin, push users to take strict precautions. Gloves, goggles, and chemical-resistant coats are standard, but I learned early that double-layered glove procedures offer real reassurance during weighing or transfer steps. Local exhaust systems prevent inhalation risk during synthesis or sampling. Incidents of skin discoloration, headaches, and mild respiratory distress have been reported among those ignoring safety. Emergency procedures stress isolation and prompt washing for spills. In regulatory terms, material safety data sheets flag it as hazardous waste, mandating careful, tagged disposal. Fire safety deserves attention too—though it won’t ignite easily, it can produce toxic fumes under thermal stress.
Application Area
Factories producing dyes, pigments, and inkjet inks run through significant quantities yearly. Its bright yellow color forms a base for more complex molecules, shifting shades by tweaking additional functional groups. In organic synthesis, it’s used to test new catalytic reactions or check the power of reducing agents. Environmental labs sometimes use it as a tracer for pollution studies because of its stability and visible color. On the education front, I recall undergraduate colorimetric determinations using this compound, which gave quick, vivid results for class demonstrations. Smaller research outfits, working on advanced optoelectronic materials, explore its interaction with polymers or as part of rare sensor platforms.
Research & Development
The molecule keeps turning up as chemists dig into better, safer colorants and functional materials. Efforts include tweaking groups around the ring to change solubility without losing color strength. Teams seek alternatives to hazardous methylation reagents, leaning on microwaves, ionic liquids, or enzyme catalysts. One research direction—practical, but overlooked—investigates lower-cost purification using membrane systems or selective crystallization. I’ve met researchers who look at this compound as a jump-off point for photonic and electronic materials—its donor-acceptor structure offers interesting charge transfer properties for sensors and organic LEDs.
Toxicity Research
Most information on health risks comes from animal and limited epidemiological studies. Exposure links to methemoglobinemia—a condition where blood can’t carry enough oxygen—especially after chronic inhalation or skin contact. Tissue accumulation and liver enzyme disruption appear in rodent studies, prompting calls for continuing medical surveillance in factories. Users face environmental risk too; it sticks around in water and soils, raising red flags for aquatic organisms. Newer work, including computational toxicology, screens derivatives for safer versions or better protective equipment standards. Ongoing regulatory reviews, especially in Europe and North America, shape handling and waste protocols.
Future Prospects
The story of N,N-dimethyl-4-nitroaniline isn’t finished. Its long record in both traditional and high-tech industries means it’ll stick around, though pressure grows to improve safety and lower waste. Alternatives for both synthesis and application are being pioneered, but the chemistry toolkit doesn’t let go of a reliable, modifiable intermediate quickly. Substitution with greener analogs depends on cost and performance, two forces that often pull in opposite directions. In academic labs, this compound can serve as a training ground for safer practice and environmental awareness. Steady research—both technical and regulatory—continues to shape how workers use, modify, and dispose of it, ensuring both progress and protection.
Beyond the Name: A Look at What It Does
Staring at the label “N,N-dimethyl-4-nitroaniline,” most folks won’t picture a packed chemistry lab. I sure didn’t, the first time I came across it while scouring resources for safer industrial dyes. This compound serves an important role as an intermediate—helping bridge complicated chemical chains—mostly in dye and pigment manufacturing. If you’ve ever wondered where the color in your favorite blue jeans or plastic containers originates, you’re looking at one of the key building blocks.
In Living Color: Dyes, Pigments, and More
Plenty of chemical products start as colorless powder, but companies who want strong, lasting yellows and oranges start with compounds like N,N-dimethyl-4-nitroaniline. In my work consulting for small-scale textile factories, I’ve watched entire batches depend on just a few grams of this stuff: a mixup at this stage, and dyes lose their strength. This compound doesn’t wind up in your t-shirt directly, but the pigments that step out of the reaction vessel would fall flat without its special chemical pattern.
Beyond textiles, the pigment industry leans on it for vibrant paints and plastics. Manufacturers synthesize what chemists call “azo dyes,” and this molecule’s nitro group helps set up the right combination of color, light-fastness, and stability. Gone are the days of colors washing out or fading in sunlight. I’ve handled products for agricultural markers—if the dye isn’t stable, farmers lose their field lines after the first rain. Compounds like this one offer a chemical backbone that delivers staying power.
The Chemistry Toolbox: Other Uses and Applications
Not every use lands on store shelves. Researchers often employ N,N-dimethyl-4-nitroaniline as an indicator, helping track reaction progress in organic chemistry labs. You see how well a reaction works, because this compound turns a vivid color when it interacts with certain substances. Its sharp response helps save time and chemical waste.
In specialty applications, some chemical manufacturers use this compound for pharmaceutical intermediates, though it’s a step or two removed from the medicine cabinet. The flexibility of its structure opens possibilities for creating molecules with new or better features. In the lab, I’ve seen students run experiments using this nitroaniline to test antioxidant capacity of natural compounds. Color changes give readouts that anyone can follow—even without fancy equipment.
Balancing Utility and Safety
Success in industry brings responsibility. Extended contact with N,N-dimethyl-4-nitroaniline can cause health risks, including toxicity through inhalation or skin exposure. Factories must stick to strict safety protocols—ventilation, gloves, and masks—to guard workers. As regulation tightens, companies and consumers push for dyes and ingredients with less environmental impact or toxicity.
Some researchers have worked toward greener dye chemistry. Finding alternatives that cut hazardous waste means everyone wins: workers, factory neighbors, and the waters downstream. Innovation starts by rethinking the very intermediates we take for granted. For students, investors, or anyone curious, the next wave of pigments may owe a debt to the lessons learned from longtime ingredients like N,N-dimethyl-4-nitroaniline.
Why Proper Storage Matters
N,N-dimethyl-4-nitroaniline isn’t one of those everyday chemicals that sits harmlessly on a shelf. It’s used in dyes, pigments, and sometimes makes its way into research labs for more specialized work. In the hands of someone who understands what it can do, it brings value. Left in the wrong spot or forgotten in the wrong container, problems start cropping up.
Too much heat, loose-fitting lids, or light pouring in from a window—these things may not seem like a big deal, but I’ve watched more than one lab scramble to clean up after a spill or a start of decomposition because storage wasn’t treated with enough respect. It’s not a scare tactic; the chemical has shown itself to be reasonably stable if you give it some basics, but ignore those rules and you’re rolling the dice.
The Essentials: Temperature, Light, and Air
Start with temperature. Shelves in a cool, dry room do the trick. Most chemists I know trust ranges from about 2°C to 8°C, like you’d find in a standard refrigerator, unless the supplier gives stricter directions. Avoid keeping it in a space that's subject to big temperature swings—sunny windowsills or laboratories close to boilers are a recipe for headache. Excess warmth encourages breakdown and can drive off volatile traces, meaning you lose both safety and the quality of your supply.
Next comes keeping the light out. This compound, like a lot of nitro aromatics, gets touchy with light. It doesn’t react well to ultraviolet rays. Use amber glass containers. I’ve seen people try to wrap clear bottles in foil, but even then, accidents happen: someone strips off the covering without thinking. Just go with the right vessel from day one.
Airtight lids aren’t an extra—they’re critical. Oxygen and moisture from the air encourage slow changes in the compound, sometimes without obvious warning. A screw-top with a good seal blocks out humidity. If you have a desiccator handy, storing the container inside adds an extra layer of insurance. Humidity control avoids clumping and keeps the product free-flowing, but also helps prevent side reactions that cut into purity or create garbage byproducts.
Labeling, Segregation, and Access
Every workplace’s got that one shelf where somebody stores a handful of orphan compounds “just for now”—and those are the bottles that get overlooked or mixed up. Clear, durable labeling matters. Put down the name, date received, and who logged it. With chemicals like this, nobody should play guessing games. Segregating it from incompatible substances is straightforward: keep it away from strong oxidizers, acids, and sources of ignition. No one appreciates finding out the hard way that materials don’t mix.
Access control isn’t about distrust. Ask anyone who’s managed a storeroom after an incident—unrestricted access raises the risks of careless handling and theft. Only trained folks who understand the hazards and proper spill clean-up should pull this compound down.
Better Storage, Fewer Headaches
Plenty of accidents could have been avoided if somebody thought ahead about safe storage. People stay healthy, supplies stay usable, and messes get kept to a minimum. Most suppliers include a sheet with all the basic rules, but the best safeguard is a culture where nobody cuts corners on things like sealing caps, labeling, and respecting storage temperature.
The Chemical in Focus
N,N-dimethyl-4-nitroaniline stands out as more than just a laboratory curiosity. This chemical, bearing both nitro and aniline groups, finds a place in dyes, pigments, and occasionally as an intermediate in chemical syntheses. Its vibrant yellow appearance hides a less-visible side—health and safety risks worth taking seriously if you find yourself exposed to it.
Human Health Concerns
Exposure to N,N-dimethyl-4-nitroaniline primarily happens through inhalation, skin contact, or accidental ingestion in lab or industrial settings. Once in the body, aromatic nitro compounds often cause problems because of how they're metabolized. Many nitroanilines break down into substances that put extra strain on the liver and kidneys. From personal experience, working alongside chemists handling these substances means respecting gloves, chemical hoods, and regular skin checks for stains or irritation.
Acute exposure sometimes irritates skin and eyes, triggering redness and itching. If inhaled, users report headaches and fatigue—a warning sign for anyone spending hours in poorly ventilated labs. Chronic exposure paints an even tougher picture. Repeated, low-level exposure over months sometimes brings on symptoms of methemoglobinemia, where the blood's ability to carry oxygen drops. Signs include blue-tinged lips or skin, dizziness, and in severe cases, unconsciousness. For people with preexisting blood disorders or respiratory conditions, the threshold for problems drops even lower.
Environmental Impact
Disposable waste containing nitroanilines often gets flagged for special handling. Research has shown that these compounds linger in the soil and water. Microbes don’t easily break down the nitro group—meaning the compound hangs around, sometimes winding up in runoff or groundwater. Fish and other aquatic life take a hit from even trace amounts, with studies noting developmental problems and impaired oxygen absorption in exposed species. Community water managers know that dye industry wastewater brings anxiety because nitroaromatic residues affect both visible pollution and the unseen, persistent toxins that stack up in rivers over time.
Regulations and Safe Handling
Manufacturers and researchers find themselves on the hook for detailed documentation and training. The U.S. Occupational Safety and Health Administration (OSHA) lists nitroanilines as hazardous materials, so companies build their safety plans with required ventilation, spill response, and medical monitoring. Europe goes a step further through the REACH framework, demanding tighter disclosure, labeling, and restrictions for persistent, bioaccumulative, and toxic substances.
Workplace best practices make a real difference. Proper storage—cool, dry, sealed containers—keeps vapors to a minimum. Ventilation systems and local exhausts go a long way in lowering airborne risks, especially when processes heat the chemical. Anyone working directly with it needs nitrile gloves, goggles, and lab coats, swapping them out after spills. Regular health monitoring means that subtle symptoms—shortness of breath or lingering headaches—get caught before they spiral.
Moving Toward Safer Choices
At universities and chemical companies, green chemistry gets more than lip service. By designing alternatives for dyes or pigments, chemists work to phase out persistent nitroanilines. Some research teams now test plant-based ingredients with similar color profiles or use biotechnology to grow safer analogs. These cleaner swaps help shrink both immediate workplace risks and the environmental footprint left behind.
Building Awareness Saves Trouble
Not everyone comes in contact with chemicals like N,N-dimethyl-4-nitroaniline, but the costs of ignoring its toxicity get high. Chemical stewardship starts with honest conversations and realistic safety steps. Protecting workers and the environment isn’t just about ticking boxes—it’s part of building trust in every lab and factory where these chemicals play a role.
A Close Look at the Structure
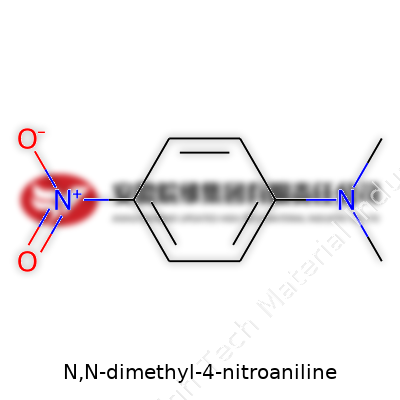
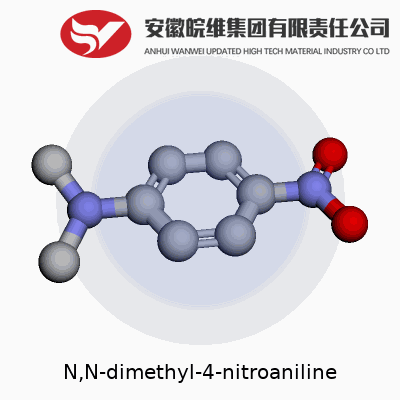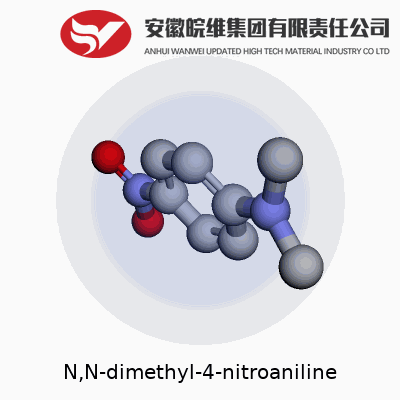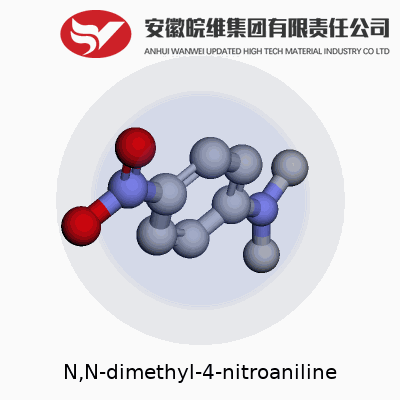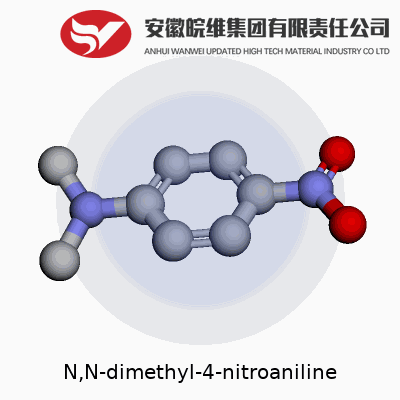
N,N-dimethyl-4-nitroaniline shows up in organic chemistry as a curious little molecule with some punchy applications. If you see the name, it breaks down like this: an aniline base (that's a benzene ring with an amino group), but in this case, the nitrogen carries two methyl groups (the "N,N-dimethyl" part). Next, a nitro group sticks to the fourth carbon on the ring. Chemically, its structure looks like this: a benzene ring with a nitro group (NO2) at the para position and two methyl groups attached to the nitrogen sitting at the amino position. It’s often drawn as C8H10N2O2.
Everyday Connections and Why It Matters
This compound might sound like something that belongs in a textbook. Growing up around a local painting business, I used to hear guys talk about “amino dyes” and why certain paints seemed to pop off the walls better than others. Colors sometimes come down to molecules like this. The nitro group gives that ring an extra jolt, changing color properties. N,N-dimethyl-4-nitroaniline has found use as a so-called “azo dye intermediate.” In other words, it helps chemists make those bright yellows and oranges you see on warning labels or playground equipment.
Beyond the hardware store, labs use this molecule for research. That double-methyl on the nitrogen and the big, electron-hungry nitro group create a unique electronic environment chemists study for reactions like nucleophilic aromatic substitution. This is a fancy way of saying swapping out little groups on the ring – and sometimes these basic concepts end up in whole new medicines or vivid synthetic pigments.
Potential Risks, Practical Thoughts
Safety with chemicals like this has always weighed on my mind – maybe it comes from helping out during high school chemistry and hearing stories of old factories where people got sick from dye exposure. The nitro-aniline family comes with a toxicity warning. Exposure can affect blood oxygen transport, leading to problems that go beyond coughing or sneezing. Handling should always include gloves, proper ventilation, and avoiding skin contact. If someone spills it, clean up has to follow good protocols, not cut corners. Respect for the material and knowing your equipment means much more than memorizing numbers on an MSDS.
The Bigger Picture: Responsible Chemistry
It’s easy to see chemistry as just memorizing names, but every compound interacts with people and places. Companies that manufacture or use N,N-dimethyl-4-nitroaniline get watched closely. Environmental guidelines push for careful waste treatment so that nitro compounds don’t show up where they shouldn’t. Back in the day, chemical runoff left scars on lakes and rivers near some plants; now, technology and regulation aim to keep these outcomes in the past.
Looking forward, chemists keep hunting for greener ways to synthesize these intermediates, using less energy or safer reagents. Students can drive that change—every time someone stops to ask, “What happens if this leaks?” or “How do I make this safer?” the field grows stronger. Whether in a textbook, on a factory floor, or mixed into an art project, compounds like N,N-dimethyl-4-nitroaniline serve as a reminder: the things built in the lab ripple outward.
Purity Matters in the Lab
N,N-dimethyl-4-nitroaniline isn’t a household name, but in chemical research and manufacturing, it’s got a reputation. Anyone who’s spent time running synthesis reactions or quality testing knows the difference between high purity and “close enough” can mean missing a useful result or ending up with a whole mess in the lab. For this compound, 97% or higher stands out as the common benchmark for purity among respected suppliers. Chemists with years at the bench often look for a certificate of analysis with every bottle, since one unpredictable impurity can ruin a batch or send a project down the wrong path.
Analytical data from Sigma-Aldrich and Thermo Fisher both show offerings around the 97%–99% purity range, with lab reports specifying the minute traces of remaining material. That’s a percentage that allows reactions to proceed smoothly, no surprises. Labs aiming for especially sensitive results or regulatory compliance sometimes pay a premium for 99% or higher, but for most applications in synthesis, dye manufacture, or as an intermediate, the 97% threshold covers the bases.
Packing Chemistry for Real-World Use
The packaging story isn’t just about glass bottles lining a storage shelf. Safety and handling always shape the choices. N,N-dimethyl-4-nitroaniline comes in amber glass bottles most commonly, with typical sizes running from 5 grams up to 500 grams. Larger consignments for industrial work might come in sealed drums, but in academic or specialty labs, the small glass bottle is king. This protects the powder from light and moisture, and with personal experience, opening a fresh, well-sealed bottle gives confidence the compound inside hasn’t degraded or clumped.
I remember buying a batch from a surplus distributor that came in a battered brown glass container, the cap loose enough to let air inside. We checked it on an NMR right away, and the compound had picked up moisture, which changed the expected reactions downstream. It cost us a week of troubleshooting. That lesson sticks: always go for sealed, tamper-evident caps. The best suppliers use leakproof closures and additional outer seals, which are details a buyer appreciates after seeing what happens otherwise.
Hazards and Real-World Decisions
This isn’t a compound for recreational chemists. N,N-dimethyl-4-nitroaniline is toxic and shows signs of being harmful if handled without the right precautions. The nitro group raises concerns about skin exposure and inhalation, and glove stains last for days. MSDS sheets from leading manufacturers set out storage below 30°C and recommend limiting its exposure to heat or sunlight, which justifies the use of thick, UV-blocking bottles.
Some suppliers send the material with extra packing: double-bagged in foil or plastic inside the glass. This can annoy anyone in a hurry, but after a spill in a shipment years ago, I saw how a second layer saves hassle and keeps hazards contained to a manageable level.
What’s Needed for Better Handling
Getting pure N,N-dimethyl-4-nitroaniline isn’t the end of the story. Labs and manufacturers can do better by insisting on transparent documentation, proper hazard labeling in multiple languages, and clear batch tracking. Purchasing managers who have dealt with mislabeled stock learn to ask ahead: is every bottle labeled with the actual batch and analysis? Is the shipping box padded well enough for bumpy transport? Solid answers to these questions save labs and workplaces time, money, and peace of mind.
Global regulations and stricter workplace safety standards have raised the bar, but that shouldn’t be a box-ticking exercise. With substances like these, practical experience matters—people who’ve worked with the stuff know the value of taking every precaution from shipment through storage, and not cutting corners just because purity numbers look impressive on paper.