Di-tert-butyl Peroxide: A Field Perspective
Historical Development
Decades ago, chemists saw di-tert-butyl peroxide as a solution to the challenge of reliable radical generation. Early studies from the 1940s tracked its use in free-radical polymerization, especially as new plastics and rubbers started to change manufacturing. Industrial demand grew in the post-war years, with capacity rising and safety improving in parallel. Methodologies shifted from small-batch work to continuous processes, making it clear that di-tert-butyl peroxide had earned its place as a backbone chemical for radical chemistry. By the late 1960s, it was everywhere from bench-top synthesis labs to sprawling polyolefin factories. Many of the classic papers that shaped peroxide chemistry came from that era, with a mix of curiosity and genuine need driving both safety improvements and technical innovation.
Product Overview
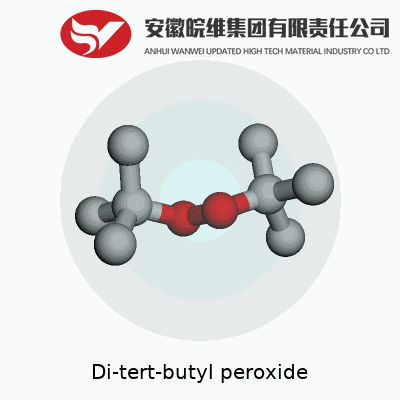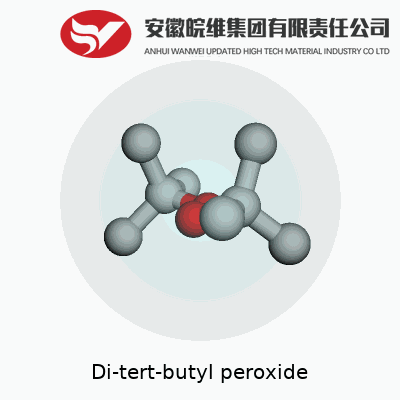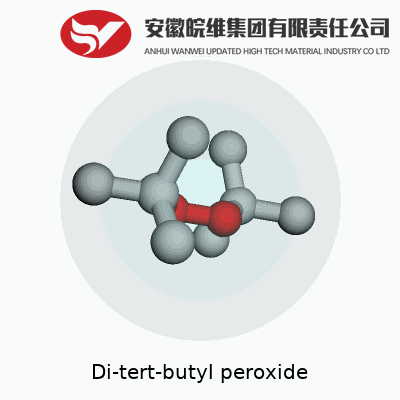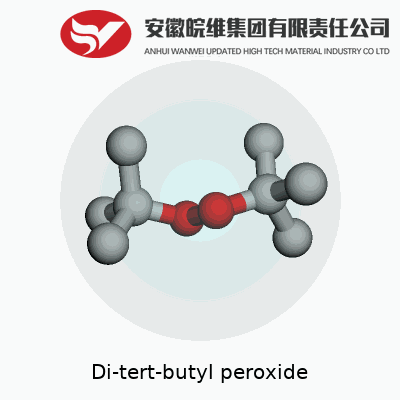
Di-tert-butyl peroxide packs a punch as an organic peroxide with a reputation for its thermal stability and predictable breakdown. Its formula, C8H18O2, reflects two bulky tert-butyl groups flanking an oxygen-oxygen bond. This molecule stands out for those who seek a consistent, reliable initiator for free-radical reactions. Not limited to just starting polymer chains, it finds roles in cross-linking, curing, and certain oxidation reactions where the reaction environment demands a persistent free-radical source. Chemists and engineers use it because of this mix of reliability and reactivity, which can give controlled results at the right temperatures.
Physical and Chemical Properties
This oily liquid brings a mild, ether-like smell to the lab. Its boiling point hovers near 111°C, with a melting point just below room temperature, so it generally stays liquid unless kept cool. The density checks in a bit lower than water, allowing it to float on top unless emulsified. What stands out is how it holds together until it hits higher temperatures, at which point it kicks into gear with a rush of exothermic decomposition. That quality allows storage under the right controls, without the quirks of more sensitive peroxides that can go off at room temperature. Handling it near open flames or sparks gets risky, which anyone using it in bulk quickly learns. Its solubility in hydrocarbons means it blends into common industrial feedstocks without phase-transfer tricks. Those designing a reaction benefit from its slow, predictable breakdown—not too rapid, not too sluggish, which makes engineering controls easier to dial in.
Technical Specifications and Labeling
Manufacturers supply di-tert-butyl peroxide with purity often exceeding 98%, and careful labeling on drums or bottles always notes flammability, storage temperatures, and emergency handling steps. Product shipments arrive with clear hazard pictograms and UN 3103 classification, as well as batch-specific certificates of analysis. Labels must warn about explosive decomposition and incompatibility with strong acids, bases, and heavy metals. In workspaces, it’s typical to see secondary containment and explicit, color-coded signage for organic peroxides. For those in operations, reading a spec sheet teaches you to watch for water content, non-volatile matter, and stability against temperature swings. These aren’t theoretical details—they link directly to safe storage and reliable processing.
Preparation Method
Chemists synthesize di-tert-butyl peroxide by reacting tert-butyl hydroperoxide with sulfuric acid in the presence of tert-butanol or isobutylene, often under controlled temperatures to encourage the right pathway. The process takes patience, as side reactions can rob efficiency, so careful adjustment of acid strength, temperature, and reactant flow rates gets the best yield. Some facilities use continuous stirred-tank reactors, and the purification step involves distillation and separation from lower-boiling impurities. These steps demand both precision and experience, since contamination with water or certain metals can degrade stability. Batch records help pin down which parameters led to higher conversion or fewer byproducts, a valuable practice when quality counts.
Chemical Reactions and Modifications
Drop di-tert-butyl peroxide into a heated reaction flask, and its bond cleaves to throw off two tert-butoxy radicals, each ready to snatch a hydrogen or initiate chain reactions. This property sits at the base of its role in synthesizing specialty polymers, especially those needing cross-linking or grafting with functional monomers. In oxidative reactions, these radicals show up as agents for selective C-H activation. In my own lab work, controlled dosing allowed smooth conversion without runaway heat. Modifying the peroxide by incorporating substituted tert-butyl groups can shift decomposition temperature, a trick sometimes used for demanding syntheses. Whether making tailored resins or altering surface properties, reaction design leans on the dependable radical punch this molecule brings.
Synonyms and Product Names
Outside of the technical literature, this chemical goes by several names—DTBP, bis(1,1-dimethylethyl) peroxide, and in some catalogues, Perkadox 16 or Vul-Cup R. Regulatory lists and databases call it di-tert-butyl peroxide. In practical settings, the abbreviation DTBP gets tossed around most, especially in production schedules or safety drills. Synonym familiarity can sidestep dangerous mix-ups; more than one close call in warehouses could have been avoided if all teams used consistent names for inventory and reporting.
Safety and Operational Standards
Many stories about accidents with organic peroxides share a theme: underestimating their hidden power. Di-tert-butyl peroxide counts as less sensitive than some cousins like benzoyl peroxide, yet complacency still spells trouble. Proper personal protective equipment—nitrile gloves, faceshields, flame-resistant lab coats—remain non-negotiable. Adequate ventilation, regular drum inspections, and limiting exposure to sunlight or heat sources go a long way. Storage in cool, shaded areas below 30°C, away from acids or rusting metal racks, avoids stability surprises. Engineering controls like explosion-proof hoods and safety showers belong near any operation handling kilos at once. Risk registers and training drills turn theoretical dangers into practical routines. Incidents taught the value of written procedures, not just oral tradition in lab groups.
Application Area
Industry uses di-tert-butyl peroxide to set the pace in modern plastics, elastomers, and wire insulation. Its radicals help tune flexibility and cross-link density in polyolefins and rubbers, important for products that need to survive decades under the hood of a car, or in power cables buried miles underground. The oil and gas sector relies on it for modifying drilling fluids and controlling polymer rheology. In specialty synthesis, chemical producers look to DTBP for selective transformations where milder agents fall short. My time in plant process optimization drove home its value in balancing reaction controllability with production economics. Small-scale fine chemical makers appreciate its reproducible effect in batch operations. This utility makes it a staple from petrochemical giants to innovation-driven specialty labs.
Research and Development
Researchers dig into di-tert-butyl peroxide for advances in polymer science and new synthetic routes. Teams publish on cleaner, greener catalytic systems that use less peroxide thanks to tailored metal catalysts, reducing waste and occupational risks. Environmental groups press for lower-release processes, and new encapsulation technologies that minimize vapor loss are emerging. Some materials scientists push for gentler radical sources, and the data from these projects push regulatory guidelines forward. Ongoing studies look at using related peroxides to modify agricultural polymers, aiming for biodegradable end products with industrial strength. Collaborative work bridges industry and academia in search of better safety testing and applications that stretch traditional boundaries. The constant push for process intensification means engineers aim for ever-tighter control and minimal byproducts.
Toxicity Research
Toxicologists have studied short- and long-term exposure, noting that DTBP’s biggest threats show up as irritation, possible organ effects, or respiratory stress if inhaled under accidental release. Animal data help frame occupational exposure limits, and industrial hygienists monitor air levels using proven detection badges and routine air sampling. Reports of chronic effects remain uncommon under proper handling, but the history of chemical industry accidents means no one lets down their guard. Lessons from near-misses led to design improvements in air handling and emergency response. Spills need quick isolation, and cleanup suits with self-contained breathing apparatuses lower personal risk. Regular safety audits look for new problems as production scales up or equipment ages.
Future Prospects
Di-tert-butyl peroxide faces both opportunity and scrutiny on the horizon. Green chemistry priorities push scientists to seek routes with fewer hazardous byproducts, and manufacturers invest in closed systems to cut emissions and waste. Regulations keep tightening, nudging plants to invest in better detection, safer storage, and more automation. Synthetic routes that regenerate or recycle peroxide byproducts have started to appear, driving down cost and improving sustainability metrics. Some of the most promising developments hinge on catalysts that unlock more value from a given dose of DTBP. The next chapter of industrial chemistry will likely keep DTBP in the toolkit, but with smarter, safer, and more environmentally friendly practices leading the way.
Unlocking the Power of Radical Chemistry
Di-tert-butyl peroxide sounds like something you’d find deep in a chemical lab—and you would, but its reach goes far beyond beakers and test tubes. In my years following the chemical industry, one thing that’s always stood out is how some of the most impactful substances tend to fly under the radar. Di-tert-butyl peroxide is one of those. This compound finds its way into places you wouldn’t expect, doing jobs that most people never see but that ripple out to make a real difference.
Pushing Polymers Further
One of the prime uses shows up in plastics. Plastics don’t just happen; they need a little nudge to start sticking together in the right way. Manufacturers add di-tert-butyl peroxide to spark polymerization, especially with tough customers like polyethylene and polypropylene. What really grabs me about this process—something that’s easy to overlook—is consistency. Factories demand the same quality every time. Small changes cost money. Di-tert-butyl peroxide hits the mark for reliability, and this makes it a staple in factories that churn out everyday items like pipes, bottles, and car parts.
Clean Combustion, Cleaner Air
Another place di-tert-butyl peroxide plays its part is in fuel. Gasoline doesn’t burn completely on its own. Incomplete combustion means more carbon monoxide and less power. Additives can make a world of difference. This compound works as an ignition improver and a cleaner-burning enhancer. I’ve seen that regulations on air quality keep getting stricter, and every little bit counts. Refineries and fuel blenders use di-tert-butyl peroxide because it helps squeeze out more efficiency from engines and, just as importantly, cuts down on tailpipe emissions. Better combustion means fewer pollutants and more miles per gallon.
Energizing the Curing Process
Over the years, I’ve learned that some of the toughest challenges in manufacturing are about time. Factories can’t wait around for products to set. This chemical speeds up the curing of resins. Boat hulls, wind turbine blades, and even some coatings on metal rely on resins that demand just the right push to harden quickly and evenly. Workers get to handle finished goods sooner, companies save time, and energy gets used more efficiently.
Handling with Care
Anyone who’s worked with di-tert-butyl peroxide will tell you it packs a punch—a very reactive chemical. There have been real accidents tied to poor handling. So, safety isn’t just a suggestion. Good training, proper storage, temperature controls—all these steps protect lives and property. The right procedures, regular inspections, and employee education go a long way in keeping accidents off the front page. In my visits to factories, attention to detail always separates the operations with records worth admiring from those that cut corners.
Looking Ahead
Growing pressure from environmental regulations means industries keep tweaking their recipes for plastics, fuels, and coatings. Over-reliance on any single substance can pinch supply. While di-tert-butyl peroxide proves its value time and time again, I think there’s room for alternatives and improved safety protocols as well. New catalysts, bio-based initiators, or even greener manufacturing techniques present some hope for reducing risks and broadening options.
Underlying all this is a simple truth: chemistry keeps the world moving. Compounds like di-tert-butyl peroxide work quietly behind the scenes, helping build, energize, and clean up the everyday products we rely on. Balancing effectiveness, safety, and sustainability will always mark the real progress in this field.
Understanding What Di-tert-butyl Peroxide Really Means for Safety
Being around chemicals has been part of my work for a long time. Di-tert-butyl peroxide looks like one of those substances that you could find in a lot of industrial processes—plastic manufacturing, specialty polymers, and sometimes even in labs where researchers try to speed up a reaction. From what I’ve seen, this compound is never just another bottle on the shelf; it comes with a list of risks that can’t be swept aside with wishful thinking.
More Than Just a Flammable Liquid
There’s no sugarcoating it: Di-tert-butyl peroxide burns easily and explodes under the right conditions. That trait alone sets off alarm bells. It doesn’t ask for a fancy ignition source. A simple spark—sometimes a static charge—is enough to start a fire. Just opening a container near a heater could cause an incident that ruins a whole line of machinery, not to mention the threat to human life. At 41°C (about 106°F), vapors start forming in earnest. In a closed space, those vapors build up quick, turning a small room into a time bomb waiting for any ignition source.
Bad News for Skin, Eyes, and Lungs
Most folks who handle chemicals start out feeling cautious, but over time habits creep in. I’ve watched people lose respect for PPE, skipping gloves or puddling a spill without a second thought. A splash of di-tert-butyl peroxide on your skin can mean burns, blistering, or at best, an uncomfortable rash. Get it in your eyes and there’s an ambulance ride in your future. Breathing the vapors—especially in unventilated spaces—tears up the nose and lungs with headaches and nausea at low doses, and much worse at higher concentrations. The American Conference of Governmental Industrial Hygienists has marked this chemical as a significant irritant, and that’s an understatement.
Explosion Hazards Make Storage a Game of Tetris
I’ve learned that if you don’t take storage seriously, you’ll get burned. Di-tert-butyl peroxide breaks down over time, especially if heat seeps in or sunlight hits the bottle. That slow breakdown releases gases, which build pressure. The next thing you know, a tiny bump or a drop could blow a cap clean off. Chemical storage rules aren’t for bureaucrats—they save lives. Separation from flammable materials and an eye on the thermometer are habits that make the difference.
How Do We Lower the Risks?
Fire, health, and explosion problems can’t get solved with half-hearted solutions. Training has to be more than a box to check. From my experience, nothing beats hands-on drills, spill clean-ups, and real-world reviews of lab layouts. Ventilation matters; not just a cracked window, but a working fume hood or an exhaust system with routine checks. Spill kits should actually have what you need—neutralizers, absorbents, and a nearby eyewash station that isn’t just for show. Anyone handling di-tert-butyl peroxide should have regular reminders about the stuff’s hazards. After all, it only takes one mistake to turn a normal day into a disaster.
Keeping an Eye on Regulations and Real World Practice
OSHA and the EPA place restrictions on how much di-tert-butyl peroxide you can store, how you transport it, and what to do with waste. It can feel bureaucratic, but every rule came from lessons learned the hard way. Factories and labs that cut corners eventually get caught—sometimes after a serious accident. Inspections and regular safety assessments keep everyone honest and sharp. Talking about chemical safety doesn’t just help comply with the law; it keeps people’s lives and livelihoods out of the emergency room.
Final Thoughts from the Floor
At the end of the day, di-tert-butyl peroxide stands as a reminder that chemical handling rewards respect and punishes neglect. Simple habits—correct storage, working ventilation, regular training—become life savers. Nobody plans on having an accident. Real safety starts by expecting one and doing everything possible so it never happens.
Getting Real About Chemical Storage
Di-tert-butyl peroxide isn’t just another lab supply you toss in the corner. This substance packs serious heat—literally and figuratively. Anyone who’s spent time around chemicals knows the risks that come with peroxides, and safety manuals weren’t written just for fun. The truth hits home after a few close calls in a lab. One memory: a mentor griping every time someone left bottles of volatile chemicals near the window. Sunlight and high temps don’t mix well with energetic molecules like these. So what’s really at stake with improper storage? Fires, explosions, lost data, damaged facilities, injuries, and the kind of “review board meeting” nobody wants.
The Science Behind Stability
Di-tert-butyl peroxide wants to break down and release oxygen, which means an energy release. At room temperature, it’s already on the edge. At higher temperatures, that risk jumps fast. Data from the National Institute for Occupational Safety and Health show the decomposition point starts north of 100°C, but you never want it to get anywhere close. The right call: store it in a refrigerator or cool room, away from direct sunlight. The storage area should always be fire-resistant.I once worked for a university that invested in explosion-proof refrigerators—those extra-thick doors and spark-resistant switches kept people less anxious. The extra money spent beats the heart attack you get from a midnight call to campus security.
Humidity, Spills, and Storing with Smarts
Moisture invites problems by attacking the container, breaking seals, or allowing corrosion. That’s a recipe for leaks and, worse, unexpected reactions. So don’t skimp on airtight containers and always keep them upright. Never store di-tert-butyl peroxide near acids, reducing agents, or heavy metals. Experience shows that careless mixing—especially during crowded, end-of-semester cleanouts—brings headaches or worse. Segregating peroxides, labeling them clearly, and keeping up-to-date records avoids confusion.A quick tip learned after a scare: don’t move these bottles more than you have to. Frequent jostling can cause unnoticed cracking or micro-leaks, which only show up when it’s too late.
Regulation and Responsibility
The Occupational Safety and Health Administration (OSHA) doesn’t play around when it comes to organic peroxides. Inspections focus on two things: temperature control and containment strategies. A real-life inspection I witnessed ended with chemical storage being reorganized from top to bottom. No more live wires nearby, no more cardboard boxes, and every bottle checked for expiration dates. Following manufacturer guidelines matters—use recommended containers and inspect them for breaks or damage.
Backing Up Good Practice with Good Training
Training isn’t a ritual; it saves lives. Every new chemist at my old workplace spent hours on peroxide safety. Practical drills—like mock spills or rush-evacuations—teach what a warning label can’t. The real test came during a minor spill, one that could’ve gone poorly. Because of those drills, the team knew how to isolate the area, ventilate the space, and deal with the container. The key: don’t wait for an accident to value training. Everyone working with di-tert-butyl peroxide benefits from hands-on safety lessons, detailed SOPs, and open communication about hazards and near-misses.
Moving Toward Safer Labs and Warehouses
Storing di-tert-butyl peroxide isn’t just a box you check on a compliance sheet. It’s about respecting every lesson learned the hard way. Manufacturers, managers, and hands-on workers keep each other safe by being vigilant, keeping protocols fresh, and sharing what they know. Strong storage practices don’t just avoid fines—they build trust from the bench to the boardroom.
Breaking Down the Formula
Di-tert-butyl peroxide shows up in labs as a clear liquid, sometimes used to trigger chemical reactions thanks to its ability to break down and release potent radicals. The formula for this compound is C8H18O2. Each molecule packs eight carbon atoms, eighteen hydrogen atoms, and two oxygen atoms. These atoms join together so that the two bulky tert-butyl groups connect through a peroxide linkage — that single oxygen-oxygen bond right in the middle.
Why the Structure Matters
Anyone who’s handled organic chemicals might recall how the shape and makeup of a molecule often decide its fate in a reaction. With di-tert-butyl peroxide, the large tert-butyl arms keep the peroxide bond tucked away, making the molecule pretty stable at room temperature compared to smaller peroxides. But heat this stuff up, and things get interesting. The O–O bond splits, spawning two tert-butoxy radicals. Those radicals make this compound a go-to initiator for polymerization and some specialty oxidation processes.
Safety and Handling
On paper, di-tert-butyl peroxide solves plenty of problems in synthetic chemistry. In practice, it demands real respect. As someone who spent time in an organic synthesis lab, I’ve seen how quick missteps with peroxides can lead to ruined experiments — or worse. Like many organic peroxides, this compound can explode under the wrong conditions. Warm temperatures, friction, or open flames all spell trouble. Eye protection, flame-resistant lab coats, and careful storage away from acids or metals turn into non-negotiables. The National Institute for Occupational Safety and Health (NIOSH) lists this chemical as an industrial hazard. Several accidents trace back to ignored safety measures, and those serve as tough reminders for new generations of chemists.
Uses: More Than Just a Lab Reagent
Beyond the test tube, di-tert-butyl peroxide finds its way into making plastics stronger and longer-lasting. Those free radicals born from its breakdown help chains of plastic snap together in the manufacturing process, especially for polystyrene or polyethylene. Some fuel blends use this compound to improve combustion efficiency, making engines burn cleaner. So, knowing the formula doesn’t just matter for balancing equations — it’s practical knowledge shaping stuff we use every day.
Concerns and Solutions for Safer Chemistry
Concerns grow as industries expand their reliance on chemical initiators. Waste and spills threaten waterways and soil. Nontoxic or less reactive alternatives are attracting research dollars and headlines. As green chemistry gathers steam, finding ways to limit or recycle hazardous byproducts from peroxide-based processes should move up on the list. Investing in closed-loop systems or digital monitoring reduces exposure risk and provides an early warning for dangerous buildups.
The Value of Chemical Literacy
It’s easy to forget the value of these formulas after the classroom. Yet, a working knowledge of compounds like di-tert-butyl peroxide makes a difference far beyond the blackboard. Chemical literacy helps keep workers safe and guides innovation toward safer and more responsible choices in chemistry. Whether mixing polymers or troubleshooting safety, understanding each atom’s role can mean the difference between progress and setbacks.
Understanding the Hazards
Di-tert-butyl peroxide sits in a class of chemicals that bring both value and risk. Most folks outside of laboratories don’t cross paths with this one, but inside research buildings and factories, it has to be respected. As an organic peroxide, it carries a real risk for fire or explosion, even with moderate shock or heat. Breathing in the fumes or getting it on your skin can land you a trip to the doctor. Handling and disposing of it calls for a game plan built on experience and discipline.
Rules That Shape Safe Disposal
For anyone working with chemicals, federal and state rules spell out the right way to dispose of hazardous stuff. The Environmental Protection Agency tags Di-tert-butyl peroxide as a hazardous waste. That’s not just a list—those rules show how big a deal careless disposal is. Pouring this into regular sinks or drains risks chemical burns, fires, or pollution. I’ve seen what can happen when labs get lazy: clogged pipes, ruined equipment, and even hospital visits.
Approaches That Actually Work
Any lab that deals with bigger volumes often uses specialized containers for organic peroxides, made from corrosion-resistant materials to block leaks. These containers have vented caps to stop pressure buildup and need to sit in cool, dry storage away from sunlight. From my experience, separating waste streams—working with small batches and not mixing peroxides with other flammable trash—pays off by cutting the risk of accidents.
The main route for final disposal runs through licensed hazardous waste handlers. These teams pack up containers, label everything with hazardous waste stickers, and haul it to facilities set up for chemical destruction. At these sites, high-temperature incinerators crank up past 1100°C to tear down organic peroxides to carbon dioxide and water. I’ve visited such sites: there’s strict recordkeeping, constant air monitoring, and layers of backup controls to avoid releases.
Why Training Matters
No system works without decent training. I once saw new lab techs almost mix different peroxide bottles. Quick action stopped a real mess. Training programs that walk staff through hands-on drills make a difference. Workers learn to spot leaking containers, handle minor spills, and use personal protective gear. Refresher classes, not just paperwork, reinforce the right habits. In high stakes work like this, it’s not about memorizing a protocol—it’s about muscle memory backed by knowledge.
Looking at Future Solutions
Plenty of research is reaching for greener chemicals that don’t leave this kind of dangerous residue behind. For now, though, strict handling and disposal will keep people safe. Tracking chemical inventory, doubling up on safety checks, and bringing in third-party audits reduce ugly surprises. Some regions push for on-site treatment units that neutralize small amounts using catalysts. These setups aren’t for everyone, but smaller research shops can rethink paying for specialized removal each time.
Responsibility Starts With Us
Any chemical, especially one as volatile as Di-tert-butyl peroxide, demands respect from start to finish. Stories in the news about fires or waste dumping often start with shortcuts or missed steps. Every person—not just the safety officer—has a role in preventing problems. With the right habits, sharp training, and support from waste professionals, safe disposal turns into a daily routine instead of a last-minute scramble.