2,5-Bis(tert-butylperoxy)-2,5-dimethylhexane: Perspectives in Chemistry and Industry
Historical Development
Chemists have explored organic peroxides for well over a century, but 2,5-Bis(tert-butylperoxy)-2,5-dimethylhexane only grabbed serious attention once polymer technology began booming after World War II. Early development focused on the challenge of producing high-energy molecules that could kick off chain reactions useful for plastics, rubbers, and synthetic fibers. Early patents traced a gradual understanding of how di-functional peroxides outperformed single-peroxide types, leading companies in both Europe and the US to scale up production throughout the 1970s and beyond. The needs of an expanding manufacturing base made this compound’s stability and reliable decomposition profile especially attractive. Industrial synthesis, safety considerations, and regulatory pressures helped refine its manufacture, raising the bar for both purity and cost-effectiveness.
Product Overview
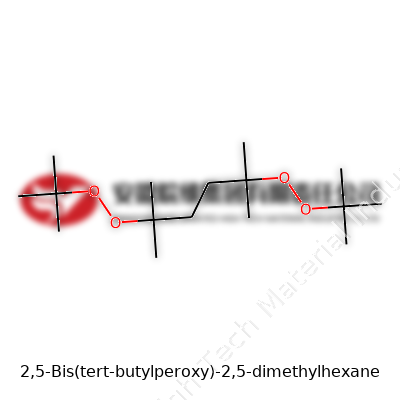
2,5-Bis(tert-butylperoxy)-2,5-dimethylhexane stands out in the toolbox of organic peroxides. Its structure, featuring two tert-butylperoxy groups attached to a branched hydrocarbon chain, delivers a solid one-two punch for applications that need precise, predictable sources of free radicals. You’ll find it in either liquid or paste form, typically stabilized with phlegmatizers to avoid runaway decomposition. The ability of this molecule to push polymerization from the inside out and crosslink polymers has set the stage for more durable plastics, making it more valuable than one might expect at first glance.
Physical & Chemical Properties
A transparent, oily liquid describes the physical presence. It doesn't just sit quietly on laboratory shelves – its boiling point hovers in high double-digit Celsius degrees, with decomposition kicking off at around 160-170°C. In the world of industrial chemistry, that window matters, offering just enough control to manage reactions but not so much that runaway heat becomes a hazard. It’s only sparingly soluble in water, so you see it blending best with hydrocarbons, esters, and other organic solvents. Its molecular formula, C16H34O4, produces a molecular weight just shy of 290 g/mol. Handling requires care due to its energetic oxygen-oxygen bonds, which can snap with force, sending free radicals flying.
Technical Specifications & Labeling
Manufacturers typically standardize 2,5-Bis(tert-butylperoxy)-2,5-dimethylhexane at high purities, usually above 95%, because impurities mess with its decomposition and performance. Technical data sheets and Material Safety Data Sheets (MSDS) spell out the details: active oxygen content, specific gravity, viscosity at various temperatures, and storage temperatures (preferably under 30°C). Labels warn of its oxidizing power, require UN classification for transport (UN 3109), and include all hazard pictograms relating to organic peroxides, flammability, and environmental risk. Regulatory agencies have forced more transparency about ingredients over the past two decades, reducing the chance of mislabeling and transport mishaps.
Preparation Method
Large-scale preparation starts with the alkylation of isobutylene to get the branched hexane backbone. Next, a careful peroxidation introduces the tert-butyl hydroperoxide. Chemists use acid catalysts and tightly control reaction temperatures. Too cold and yields drag; too hot and the product can decompose violently, endangering workers. I’ve talked with polymer plant operators who credit their process safety systems as the real unsung heroes of this synthesis. Filtration and vacuum distillation polish the product, squeezing out residual reactants that could destabilize the final compound. Reliable production lines avoid metal contamination, since transition metals can decompose peroxides and spark unplanned reactions.
Chemical Reactions & Modifications
In polymer plants, 2,5-Bis(tert-butylperoxy)-2,5-dimethylhexane finds its groove by generating free radicals that crack open monomers or crosslink polymer chains under heat. Its symmetrical structure means both ends can kick off reactions, doubling up on activity. This bifunctional setup helps create resilient, thermoset plastics and enhance the toughness of elastomers. Chemists have explored modifications by changing the alkyl substituents or adding stabilizers for tailored reactivity, but they circle back to this hexane backbone for its balance of performance and manageable decomposition. You’ll see small-scale research investigating copolymer blends or introducing polar groups, but the original remains the workhorse for most industrial systems.
Synonyms & Product Names
Not everyone calls it by its full IUPAC name. The chemical supply world shortens it to “2,5-Di-tert-butylperoxy-2,5-dimethylhexane,” among other variations: “Trigonox 101,” “Perkadox 14,” or “DTBPDH.” Each supplier throws its own branding on the bottle. Chemists ordering the stuff need to double-check CAS numbers (78-63-7) and compare specification sheets, since regional names and minor formulas can lead to expensive mis-orders. Swapping brand names between vendors demands a careful look at handling recommendations, because even inert fillers and stabilizers can change reactivity in a batch.
Safety & Operational Standards
Every operator I’ve met understands the nervous respect these peroxides command. Strict storage rules govern how much you can keep in one place, with temperature monitors and separated, well-ventilated drum storage. Personal protective equipment becomes non-negotiable; face shields, gloves, and flame-resistant lab coats make the difference. OSHA, the European Chemicals Agency, and China’s MEE lay out clear rules: real-time temperature logging, dedicated fire response systems, and container earthing. Human error remains the biggest risk, and training reduces mistakes with mixing, measuring, or disposing of leftover material. I’ve seen what happens when corners get cut: near-misses at unloading bays, drum bulges after warm weekends, and close calls with ungrounded tools.
Application Area
The world keeps leaning on plastics, especially for automotive, electrical, and construction uses, and that means constant demand for efficient crosslinking agents. This hexane-based peroxide steps up where regular initiators don’t cut it. Its ability to balance a mild decomposition profile with strong free radical output helps produce wire coatings, HDPE pipes, insulation foams, gaskets, and tire components. Its dual-function approach means manufacturers can reach higher cure depths and improved weathering resistance in finished goods. Not just a polymer story, this compound also finds secondary uses for controlled degradation in recycling, adhesive formulations, and specialty resin synthesis. I’ve watched R&D teams find launch pads for new applications by tweaking concentrations and concocting new blends, sometimes with results that look far removed from the molecule’s original purpose.
Research & Development
Innovation teams keep pushing this molecule’s limits. Some focus on reducing decomposition temperatures to boost its use in sensitive matrixes. Others tackle the ecological footprint, searching for ‘greener’ reaction pathways or safer stabilizers. Recently, collaborations between universities and industry have mapped the full breakdown path, tracking every radical and byproduct. This research set the stage for stricter emissions controls and strategies for reclaiming spent material instead of dumping it. There’s increasing interest in using it for 3D printed polymers, seeking more predictable curing in additive manufacturing. Every advancement depends on a delicate trade-off between reactivity, safety, and lifecycle cost.
Toxicity Research
Few researchers relax around organic peroxides, knowing their decomposition products and direct exposure risks. Studies show this molecule irritates skin and eyes, and repeated long-term inhalation leads to respiratory trouble in exposed workers. Animal studies point to moderate oral toxicity, but the real concern sits with explosion risk and the potential for fires in cramped storerooms or transport containers. Regulatory toxicologists track how the molecule breaks down, particularly looking for tert-butanol and other volatile organics downstream. There’s still work to do on mapping low-level, long-duration environmental impact. Most workplace incidents stem from improper mixing or accidental spills, and these lessons feed into better training and safety audits.
Future Prospects
Looking ahead, the pressures of ESG standards and consumer demand for safer chemicals push producers to refine processes even more. Automation and digital monitoring look set to cut human error, and chemists continue probing the high ground for a version with even lower potential for mishaps. Another big growth area stands at the intersection with renewable resource feedstocks—imagine using bio-based precursors or improving end-of-life management. Research teams have started exploring peroxides with switched-up alkyl chains or next-generation stabilizers, but until those go fully mainstream, 2,5-Bis(tert-butylperoxy)-2,5-dimethylhexane stays in the driver’s seat. Each batch, every safety audit, and ongoing tweaks to supply chains all play into a future where performance and responsibility walk hand-in-hand, whether in plastics now or in materials we haven’t yet dreamed up.
Walking Through the World of Plastics
Plastics show up everywhere, not just in shopping bags or water bottles but under car hoods, in home appliances, even in the soles of your sneakers. Digging deeper into what shapes and toughens these plastics, I came across some interesting ingredients. 2,5-Bis(tert-butylperoxy)-2,5-dimethylhexane—quite a mouthful—stands out because factories rely on it to make plastics and rubber products perform a lot better than they would otherwise.
The Power of Peroxides in Manufacturing
Factories use this chemical mostly as a crosslinking agent. Think of crosslinking as the process that ties all the loose ends together in a polymer chain, making materials much tougher. It’s like the difference between cooked spaghetti (wobbly, soft) and baked ziti (stick-together firmness). The crosslinked version means materials can take more heat, stress, and bending without breaking apart. That makes it a favorite in the production of things like polyethylene and ethylene-vinyl acetate (EVA) foam. Without this reaction, the soles on running shoes or the insulation on electrical wires would crack or tear too easily.
Inside Factories: Safety and Reliability First
Over the years, I have toured plastic production plants and watched the focus on safety when working with chemicals like this one. Production teams keep chemicals like 2,5-Bis(tert-butylperoxy)-2,5-dimethylhexane under tight control. Since it’s a peroxide, it packs a punch—making it essential for getting stubborn plastics to harden up but also requiring careful storage and handling. I have seen how crews use gloves and full face shields, even for routine work. No one wants an unplanned reaction, especially since this peroxide breaks down and releases energy if mishandled.
Making Products that Last
What stands out about this chemical is how it helps turn soft, pliable plastic into materials ready for rough use. The foam underfoot in gym flooring, the coating that wraps around car cables, the insulation for buried wires—products built with durability in mind. These products last longer before breaking down or turning brittle, thanks to the crosslinking brought about by peroxides like this one. Polyethylene pipes made using this chemistry continue to hold up under pressure, buried for years in municipal systems.
Addressing Concerns and Looking for Alternatives
People working near industrial chemicals always ask the same thing: how safe is it? Regulators have guidelines in place because peroxides can be risky if not used correctly. Companies set up closed systems, so emissions aren’t released into the workspace. Research continues into safer compounds or reaction methods; the industry keeps searching for new catalysts that offer the same reliability but lower health risks. Bio-based solutions get a lot of buzz, but so far, nothing matches the performance for certain industrial uses.
Choosing the Responsible Path
It comes down to balancing performance with responsibility. Factories want products that don’t fail in the field but also want to protect their workers and local communities. Every step, from shipping to storage to production, gets evaluated to prevent mistakes. It’s a daily job—improving technology, auditing safety steps, and updating rules to reflect the latest science. The plastics in your life last longer and stay safer because of that chemical and the people who treat its power with respect.
Respect the Risk Before Anything Else
I remember walking into my first university lab. The instructor didn't just hand out safety goggles—she drilled into us the idea that mistakes with chemicals do real harm. All those “small” spills, ignored smells, and unlabeled beakers set up problems that ripple beyond one person or one day at work. The sooner you understand the real risks tied to a specific chemical, the safer everyone stays. An everyday example—bleach, a chemical almost all of us keep at home. Mix it with ammonia, and you’re facing toxic gas. Understanding exactly what you’re handling doesn’t just matter in industry, it starts at home.
Personal Protective Equipment Should Never Be an Afterthought
You always see the splash of bright gloves and lab coats in photos, but nothing replaces making sure you’ve got dependable goggles, gloves that don’t split every hour, and that you watch for leaks or tears. Some chemicals hit your lungs quickly—think about strong acids or solvents—so a mask is not a suggestion, it’s a responsibility. A face shield helps when there’s risk of flying debris or splashing. Regularly check gear for cracks, especially on goggles and gloves after repeat use.
Labeling and Clear Storage Matter
It seems obvious, but it’s easier than you think to leave a bottle out or skip writing a label during clean-up. That single shortcut leads to the wrong person grabbing the wrong substance. The best solution I’ve seen is color-coded lab tape with bold, legible names—no abbreviations, no mysteries. For storage, isolation prevents dangerous reactions—never put acids near bases, and never store flammable solvents near anything with a heating element. If you keep a spill kit nearby, you’ll stop a small mess from turning into a full-blown evacuation.
Ventilation Saves More Than Just Comfort
Old buildings with locked windows—those make me nervous in labs. Strong chemicals build up in still air, ready to hurt lungs and eyes. Always open the fume hood, and never use one for storage. Strong fans pulling air away from your workspace aren’t about comfort—they turn a dangerous job into something much safer. If the air smells wrong, trust your sense—step away and call in help. Regular checks of HVAC filters and monitoring for leaks or blocked vents keep everyone safer for the long run.
Emergency Plans Separate Small Problems from Real Disasters
I’ve seen panic set in when someone spills acetone or breaks a mercury thermometer. With the right training and a calm voice, these events stay small. You need to know the route to the emergency shower, where the eyewash station stands, and who you call in case of exposure. Posting a clear step-by-step guide on the wall—visible, not tucked in a drawer—sets everyone up for quick action. During drills, pay attention, because the panic you feel during practice is nothing compared to when things go wrong for real.
Continuous Learning and Accountability
Training never ends. Chemical safety changes as new research comes out and as people make mistakes and learn from them. Everyone should share close calls—embarrassment costs less than a burnt hand. Industry figures suggest that places with regular safety reviews and open reporting see far fewer serious injuries. Keep up with updates from agencies like OSHA, and look to the Material Safety Data Sheets for clear action guides on specific risks.
Safety around chemicals demands attention, right equipment, and the courage to do things right every time.Why Storage Conditions Matter
Every process that depends on chemical ingredients benefits from paying close attention to how those compounds spend their time on the shelf. Many people hear the name 2,5-Bis(tert-butylperoxy)-2,5-dimethylhexane, often known as Trigonox 101, and think only of its usefulness as an initiator in plastics and rubber. Yet, safety and stability begin long before the first batch enters production lines. Missteps in chemical storage can set the stage for costly mistakes, injuries, or fires.
The Crucial Details: What This Compound Needs
To understand why storage conditions matter, I remember walking into a facility and seeing containers arranged for convenience, not caution. Too often, someone assumes all peroxides behave the same way, but this chemical has special demands. Its molecular structure packs a punch—those peroxy bonds react quickly to heat and can break down with alarming results if left unchecked.
Industry guidelines recommend keeping 2,5-Bis(tert-butylperoxy)-2,5-dimethylhexane in cool, dry, and well-ventilated spaces, usually at temperatures below 30°C (86°F). I’ve seen temperature logs help prevent disasters. Placing this chemical in a warehouse corner where air doesn’t circulate and the sun bears down through a window has led to containers sweating or, in one extreme case, a near miss after a string of heat waves. Peroxides do not handle warmth with grace; their decomposition risk soars if temperatures climb, producing gases that pressurize containers or fuel fires.
Humidity matters too. Moisture creeping into packaging triggers unwanted reactions, which can foul a whole shipment or threaten worker safety. Even minor leaks make a difference with this compound. I’ve watched workers with careful eyes swap out compromised seals before problems started. Good air flow helps to disperse any vapors—a practical, everyday detail that gets lost in technical manuals but cannot be overstated for keeping people and property safe.
Isolation from other chemicals ranks high on the priority list. This is not a substance to park beside acids, bases, or metal powders. In one real scenario, a facility failed to keep a separating wall between incompatible materials, and a small spill led to a tense afternoon for everyone in the building. Segregated storage—using clearly marked shelves or even separate rooms—doesn’t just check off a regulatory box. It provides real insurance against chain reactions and headaches down the line.
Supporting Safety with Best Practices
Trained staff anchor a culture of safety, more than any posted sign or sticker. Frequent reviews of handling protocols help reduce risky shortcuts, especially with turnover on the floor. Involving local fire authorities and emergency responders in planning drills shows what happens when theory meets reality. Data from the National Fire Protection Association (NFPA) points to a lower incidence of chemical incidents in sites that review safety protocols at least twice a year.
Packaging holds its own importance; only containers made from compatible, resistant materials stand up to peroxides. In my experience, it pays to invest in containers with pressure relief features, especially during seasonal swings in temperature. Clear hazard labeling and up-to-date Material Safety Data Sheets (MSDS) make everyone part of the safety team, not just the chemical manager.
Moving Forward with Confidence
Reliable safety around 2,5-Bis(tert-butylperoxy)-2,5-dimethylhexane storage comes from a blend of vigilance, planning, and respect for the compound’s sensitivity. Creating reliable checklists and sticking with a maintenance calendar does more than keep regulators happy—it becomes an everyday commitment to health, business, and future innovation. Keeping this compound stable and ready for work rests on a foundation built one careful choice at a time.
Getting Specific: The Language of Chemistry
In chemistry class, I remember staring at long strings of letters and numbers—C6H12O6, CH3COOH—and thinking, how could this cluster of symbols mean so much? Over time, it became clear that the formula isn’t just a bunch of characters, but a shortcut to understanding everything about a substance. The chemical formula tells you which atoms link up to build the compound, how many of each you’ll find, and hints at how the whole thing behaves. The structure hands you even more: it maps the way atoms fit together, forming chains, rings, or complex networks.
Small Changes, Big Differences
Once you look past the letters and start examining the arrangements, you see how a simple flip in structure can change a compound’s identity. Take glucose and fructose—both C6H12O6 on paper. Their atoms connect differently, so they taste different, act different in the body, and show up in food in unique ways. This isn’t academic trivia. Understanding these differences means safer drugs, smarter engineering of materials, and healthier food choices. It keeps industries honest too, especially in pharmaceuticals and food science, where small tweaks in structure separate a cough remedy from a banned substance.
Formula and Structure: Front Line in Research and Industry
Any scientist or quality control chemist will tell you—knowing the exact makeup of a compound underpins everything they do. Misreading a structure can send a new drug back to the drawing board, or worse, trigger side effects nobody saw coming. I’ve seen everything from cleaning products to beauty creams hinge on accurate chemical identification. A wrong call doesn’t just cost time and money; it risks health and safety.
Publishing research? Reviewers make a habit of double-checking both formula and structure, holding the work to a high standard. This careful attention isn’t a gatekeeping tactic; it stops counterfeit products and protects consumers from unsafe chemicals. Regulators like the Food and Drug Administration don’t just want a formula—they want to see diagrams, chromatography results, and repeatable experiments.
Technology’s Role in Getting Things Right
Tools like nuclear magnetic resonance (NMR) and mass spectrometry let chemists look deeper, confirming that what’s on the bottle matches what’s inside. These instruments don’t just spit out spectra for fun—they help spot impurities, reveal structural surprises, and build trust between laboratory and marketplace. As technology keeps evolving, so does the accuracy of chemical identification. It feels good to know that a medicine passed dozens of technical hurdles before landing on a pharmacy shelf.
Pushing for Transparency and Clear Labeling
If you’ve ever shopped for vitamins or checked the back of a cleaning product, you might have wondered what “inactive ingredients” really are. Industry should move toward full transparency, with detailed chemical names and diagrams where it matters. This empowers those with allergies or sensitivities to avoid trouble, and it gives professionals the info needed for emergency care or product recalls.
Education: Turning Mysteries Into Tools
By making structure and formula a core part of science education and workplace training, people grow more comfortable with chemistry. It pays off in public safety, better products, and stronger innovation. The clearer the labels and teaching, the less room for costly mistakes and confusion.
Understanding What We’re Dealing With
2,5-Bis(tert-butylperoxy)-2,5-dimethylhexane isn’t a chemical that gets used lightly. It plays a big part in making plastics tough and flexible, especially in places like automotive plants and industries working on insulation. At its core, this compound acts as an organic peroxide, which carries some real safety concerns. Even a small mistake with leftover material or residue can lead to serious fires or explosions. As someone who’s worked in research labs and seen the aftermath of hazardous chemical mistakes, I can say these risks matter both for workers and for the larger community.
Dangers You Can’t Sweep Under the Rug
This chemical reacts unpredictably to heat, shock, and friction. Storage and disposal without careful routines open the door to disaster. Spills don’t just threaten the building but also the air and water outside, since these peroxides can break down into things that cause harm. According to safety data sheets and first-hand accounts, fire departments treat these substances with careful routines for a reason. These aren’t the sorts of chemicals that get poured down the drain or tossed with everyday trash.
Professional Disposal—Not Just a Suggestion
Leaving hazardous waste to chance invites accidents and lasting contamination. Companies and labs with small amounts on hand need to call in certified hazardous waste handlers. These handlers have special trucks, containers, and gear. They also have clear routes for shipping material offsite to facilities built for dealing with stuff that could blow up or catch fire if mishandled. Once it reaches a treatment center, trained crews inspect containers for leaks and make sure everything stays stable. Then, they destroy the peroxide using controlled high-temperature incineration, which breaks down the molecules safely and keeps any byproducts from harming the neighborhood or environment.
Why Incineration Gets Chosen
Incineration doesn’t leave much behind. Temperatures soaring past typical flames break all the bonds in the chemical, leaving little beyond carbon dioxide, water vapor, and a few trace minerals. Modern incinerators catch almost every fume, cutting out most risk to the air. With this system, hazardous peroxides don’t get a second chance to react or leak out to landfills, rivers, or the air you breathe.
Hazardous Waste Laws Matter More Than You Think
Regulatory agencies like the EPA call the shots here, and for good reason. There’s a long list of waste codes and governmental rules set down for organic peroxides, drawn from a half-century of bad accidents and environmental losses. Fines and penalties never fix the health impacts of bad disposal, but they do push companies to toe the line. Local agencies track hazardous waste from cradle to grave, stamping every shipment with paperwork and details about where it ends up.
Training Workers—the Hidden Step
Many problems start with someone who didn’t get the right training. Regular, hands-on safety sessions make all the difference. In places I’ve worked, the best lab managers put everyone through yearly drills on spill response and storage routines. Proper signs, spill kits, and chemical logs take a bit of time and focus, but they’ve kept dozens of workers safe—and kept businesses running smoothly, without a visit from the fire marshal or worse.
Taking Responsibility for the Chemicals We Use
While disposal companies and government agencies lay out guidance, the daily responsibility falls on the shoulders of those using or handling 2,5-Bis(tert-butylperoxy)-2,5-dimethylhexane. If a drum goes out the door, someone signs a manifest and confirms it got there safely. Nobody likes the paperwork, but cutting corners leads to damage nobody can shrug off. It’s about protecting the people in the lab, the janitor mopping the floors, and kids playing by the river downstream. Safety beats shortcuts, every time.